| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carbonic anhydrase 12 |
|---|
| Ligand | BDBM50228328 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_469185 (CHEMBL932210) |
|---|
| Ki | 2.8±n/a nM |
|---|
| Citation |  Rami, M; Winum, JY; Innocenti, A; Montero, JL; Scozzafava, A; Supuran, CT Carbonic anhydrase inhibitors: copper(II) complexes of polyamino-polycarboxylamido aromatic/heterocyclic sulfonamides are very potent inhibitors of the tumor-associated isoforms IX and XII. Bioorg Med Chem Lett18:836-41 (2008) [PubMed] Article Rami, M; Winum, JY; Innocenti, A; Montero, JL; Scozzafava, A; Supuran, CT Carbonic anhydrase inhibitors: copper(II) complexes of polyamino-polycarboxylamido aromatic/heterocyclic sulfonamides are very potent inhibitors of the tumor-associated isoforms IX and XII. Bioorg Med Chem Lett18:836-41 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carbonic anhydrase 12 |
|---|
| Name: | Carbonic anhydrase 12 |
|---|
| Synonyms: | CA-XII | CA12 | CAH12_HUMAN | Carbonate dehydratase XII | Carbonic anhydrase | Carbonic anhydrase 12 (CA XII) | Carbonic anhydrase XII | Carbonic anhydrase XII (CA XII) | Carbonic anhydrase XII (CAXII) | Tumor antigen HOM-RCC-3.1.3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 39456.00 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Catalytic domain of human cloned isozyme was used in the assay |
|---|
| Residue: | 354 |
|---|
| Sequence: | MPRRSLHAAAVLLLVILKEQPSSPAPVNGSKWTYFGPDGENSWSKKYPSCGGLLQSPIDL
HSDILQYDASLTPLEFQGYNLSANKQFLLTNNGHSVKLNLPSDMHIQGLQSRYSATQLHL
HWGNPNDPHGSEHTVSGQHFAAELHIVHYNSDLYPDASTASNKSEGLAVLAVLIEMGSFN
PSYDKIFSHLQHVKYKGQEAFVPGFNIEELLPERTAEYYRYRGSLTTPPCNPTVLWTVFR
NPVQISQEQLLALETALYCTHMDDPSPREMINNFRQVQKFDERLVYTSFSQVQVCTAAGL
SLGIILSLALAGILGICIVVVVSIWLFRRKSIKKGDNKGVIYKPATKMETEAHA
|
|
|
|---|
| BDBM50228328 |
|---|
| n/a |
|---|
| Name | BDBM50228328 |
|---|
| Synonyms: | ([2-(Carboxymethyl-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-ethyl]-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-acetic acid; compound with Zn complex | ([2-(carboxymethyl-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-ethyl]-{[2-(4-sulfamoyl-phenyl)-ethylcarbamoyl]-methyl}-amino)-acetic acid | CHEMBL1795054 | CHEMBL34526 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H36N6O10S2 |
|---|
| Mol. Mass. | 656.728 |
|---|
| SMILES | NS(=O)(=O)c1ccc(CCNC(=O)CN(CCN(CC(O)=O)CC(=O)NCCc2ccc(cc2)S(N)(=O)=O)CC(O)=O)cc1 |
|---|
| Structure | 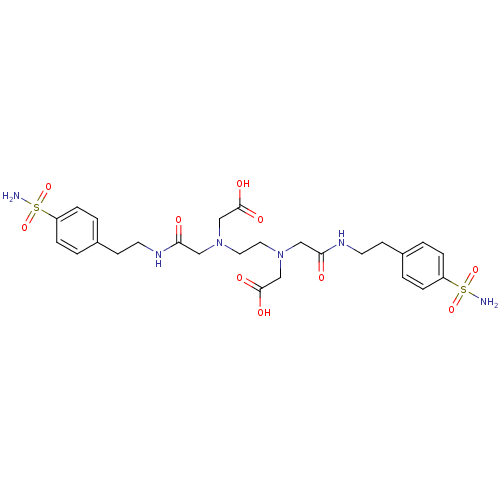
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Rami, M; Winum, JY; Innocenti, A; Montero, JL; Scozzafava, A; Supuran, CT Carbonic anhydrase inhibitors: copper(II) complexes of polyamino-polycarboxylamido aromatic/heterocyclic sulfonamides are very potent inhibitors of the tumor-associated isoforms IX and XII. Bioorg Med Chem Lett18:836-41 (2008) [PubMed] Article
Rami, M; Winum, JY; Innocenti, A; Montero, JL; Scozzafava, A; Supuran, CT Carbonic anhydrase inhibitors: copper(II) complexes of polyamino-polycarboxylamido aromatic/heterocyclic sulfonamides are very potent inhibitors of the tumor-associated isoforms IX and XII. Bioorg Med Chem Lett18:836-41 (2008) [PubMed] Article