| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50100418 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_479909 (CHEMBL923559) |
|---|
| Ki | 1660±n/a nM |
|---|
| Citation |  Melman, A; Gao, ZG; Kumar, D; Wan, TC; Gizewski, E; Auchampach, JA; Jacobson, KA Design of (N)-methanocarba adenosine 5'-uronamides as species-independent A3 receptor-selective agonists. Bioorg Med Chem Lett18:2813-9 (2008) [PubMed] Article Melman, A; Gao, ZG; Kumar, D; Wan, TC; Gizewski, E; Auchampach, JA; Jacobson, KA Design of (N)-methanocarba adenosine 5'-uronamides as species-independent A3 receptor-selective agonists. Bioorg Med Chem Lett18:2813-9 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | AA2AR_RAT | ADENOSINE A2a | Adenosine A2 receptor | Adenosine A2a receptor (A2a) | Adenosine Receptors A2a (A2a) | Adenosine receptor A2a and A3 | Adenosine receptors A2a | Adora2a | Rat striatal adenosine A2a receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 45015.65 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat A2A receptors expressed in CHO cells. |
|---|
| Residue: | 410 |
|---|
| Sequence: | MGSSVYITVELAIAVLAILGNVLVCWAVWINSNLQNVTNFFVVSLAAADIAVGVLAIPFA
ITISTGFCAACHGCLFFACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGVRAKG
IIAICWVLSFAIGLTPMLGWNNCSQKDGNSTKTCGEGRVTCLFEDVVPMNYMVYYNFFAF
VLLPLLLMLAIYLRIFLAARRQLKQMESQPLPGERTRSTLQKEVHAAKSLAIIVGLFALC
WLPLHIINCFTFFCSTCRHAPPWLMYLAIILSHSNSVVNPFIYAYRIREFRQTFRKIIRT
HVLRRQEPFQAGGSSAWALAAHSTEGEQVSLRLNGHPLGVWANGSATHSGRRPNGYTLGL
GGGGSAQGSPRDVELPTQERQEGQEHPGLRGHLVQARVGASSWSSEFAPS
|
|
|
|---|
| BDBM50100418 |
|---|
| n/a |
|---|
| Name | BDBM50100418 |
|---|
| Synonyms: | (1S,2R,3S,4R,5S)-4-(6-(3-iodobenzylamino)-2-chloro-9H-purin-9-yl)-2,3-dihydroxy-N-methylbicyclo[3.1.0]hexane-1-carboxamide | (1S,2R,3S,4R,5S)-4-[2-Chloro-6-(3-iodo-benzylamino)-purin-9-yl]-2,3-dihydroxy-bicyclo[3.1.0]hexane-1-carboxylic acid methylamide | 1N-methyl-4-[2-chloro-6-(3-iodobenzylamino)-9H-9-purinyl]-2,3-dihydroxy-(1S,2R,3S,4R,5S)-bicyclo[3.1.0]hexane-1-carboxamide | CHEMBL27376 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H20ClIN6O3 |
|---|
| Mol. Mass. | 554.769 |
|---|
| SMILES | CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(I)c3)nc(Cl)nc12 |r| |
|---|
| Structure | 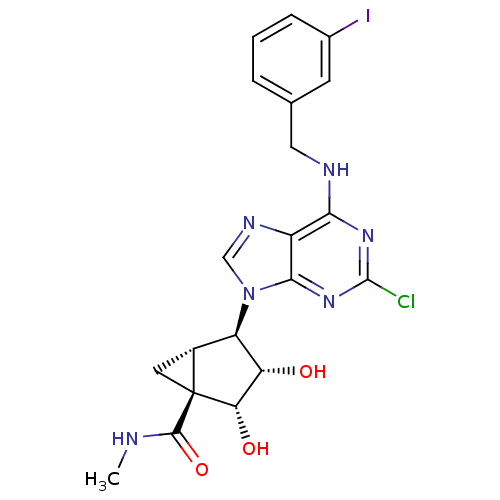
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Melman, A; Gao, ZG; Kumar, D; Wan, TC; Gizewski, E; Auchampach, JA; Jacobson, KA Design of (N)-methanocarba adenosine 5'-uronamides as species-independent A3 receptor-selective agonists. Bioorg Med Chem Lett18:2813-9 (2008) [PubMed] Article
Melman, A; Gao, ZG; Kumar, D; Wan, TC; Gizewski, E; Auchampach, JA; Jacobson, KA Design of (N)-methanocarba adenosine 5'-uronamides as species-independent A3 receptor-selective agonists. Bioorg Med Chem Lett18:2813-9 (2008) [PubMed] Article