 Found 2735 hits with Last Name = 'wan' and Initial = 'tc'
Found 2735 hits with Last Name = 'wan' and Initial = 'tc' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Homo sapiens (Human)) | BDBM19023

(1-(4-methoxyphenyl)-7-oxo-6-[4-(2-oxopiperidin-1-y...)Show SMILES COc1ccc(cc1)-n1nc(C(N)=O)c2CCN(C(=O)c12)c1ccc(cc1)N1CCCCC1=O Show InChI InChI=1S/C25H25N5O4/c1-34-19-11-9-18(10-12-19)30-23-20(22(27-30)24(26)32)13-15-29(25(23)33)17-7-5-16(6-8-17)28-14-3-2-4-21(28)31/h5-12H,2-4,13-15H2,1H3,(H2,26,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Adenosine receptor A3
(Mus musculus) | BDBM50118812
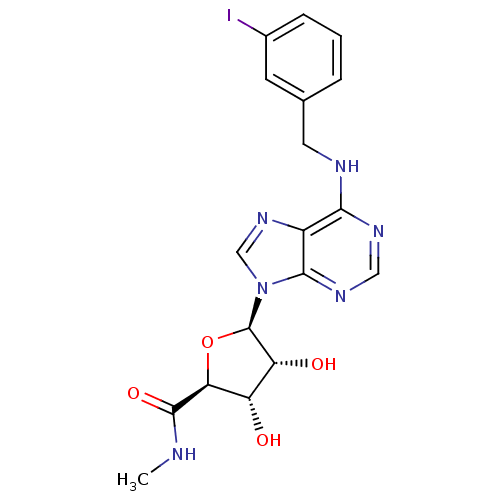
((2S,3S,4R,5R)-3,4-Dihydroxy-5-[6-(3-iodo-benzylami...)Show SMILES CNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(NCc3cccc(I)c3)ncnc12 |r| Show InChI InChI=1S/C18H19IN6O4/c1-20-17(28)14-12(26)13(27)18(29-14)25-8-24-11-15(22-7-23-16(11)25)21-6-9-3-2-4-10(19)5-9/h2-5,7-8,12-14,18,26-27H,6H2,1H3,(H,20,28)(H,21,22,23)/t12-,13+,14-,18+/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [125I]N6-(-amino-3-iodobenzyl)adenosine-5'-N-methyl-uronamide from mouse recombinant adenosine A3 receptor expressed in HEK293 cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Mus musculus) | BDBM21221
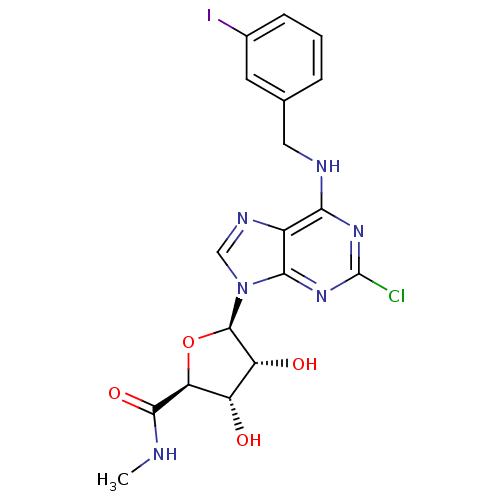
((2S,3S,4R,5R)-5-(2-chloro-6-{[(3-iodophenyl)methyl...)Show SMILES CNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(NCc3cccc(I)c3)nc(Cl)nc12 Show InChI InChI=1S/C18H18ClIN6O4/c1-21-16(29)13-11(27)12(28)17(30-13)26-7-23-10-14(24-18(19)25-15(10)26)22-6-8-3-2-4-9(20)5-8/h2-5,7,11-13,17,27-28H,6H2,1H3,(H,21,29)(H,22,24,25)/t11-,12+,13-,17+/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [125I]N6-(-amino-3-iodobenzyl)adenosine-5'-N-methyl-uronamide from mouse recombinant adenosine A3 receptor expressed in HEK293 cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12676

(1-(3-Aminobenzisoxazol-5-yl)-3-trifluoromethyl-N-[...)Show SMILES CN(C)Cc1nccn1-c1ccc(NC(=O)c2cc(nn2-c2ccc3onc(N)c3c2)C(F)(F)F)c(F)c1 Show InChI InChI=1S/C24H20F4N8O2/c1-34(2)12-21-30-7-8-35(21)13-3-5-17(16(25)10-13)31-23(37)18-11-20(24(26,27)28)32-36(18)14-4-6-19-15(9-14)22(29)33-38-19/h3-11H,12H2,1-2H3,(H2,29,33)(H,31,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218305

(3-chloro-N-((1S,2R)-2-(4-(2-oxopyridin-1(2H)-yl)be...)Show SMILES Clc1c[nH]c2cc(ccc12)C(=O)N[C@@H]1[C@@H](Cc2ccccc12)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C30H23ClN4O3/c31-24-17-32-25-16-20(10-13-23(24)25)30(38)34-28-22-6-2-1-5-19(22)15-26(28)33-29(37)18-8-11-21(12-9-18)35-14-4-3-7-27(35)36/h1-14,16-17,26,28,32H,15H2,(H,33,37)(H,34,38)/t26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218298
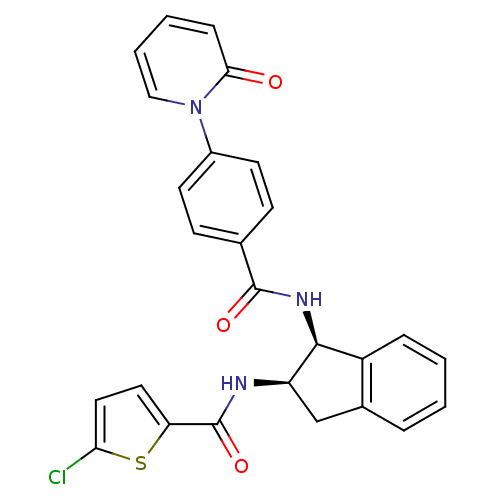
(5-chloro-N-((1S,2R)-1-(4-(2-oxopyridin-1(2H)-yl)be...)Show SMILES Clc1ccc(s1)C(=O)N[C@@H]1Cc2ccccc2[C@@H]1NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C26H20ClN3O3S/c27-22-13-12-21(34-22)26(33)28-20-15-17-5-1-2-6-19(17)24(20)29-25(32)16-8-10-18(11-9-16)30-14-4-3-7-23(30)31/h1-14,20,24H,15H2,(H,28,33)(H,29,32)/t20-,24+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50163020
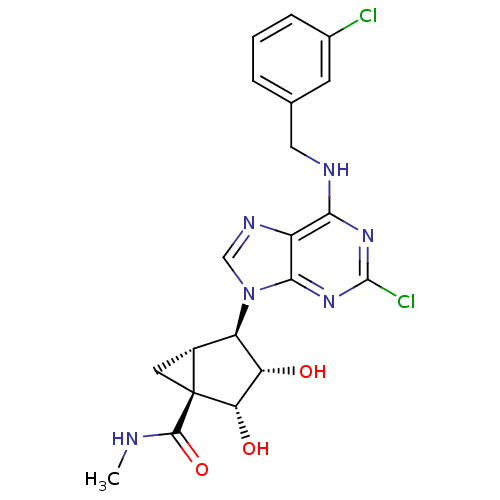
((1S,2R,3S,4R,5S)-4-(6-(3-chlorobenzylamino)-2-chlo...)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Cl)c3)nc(Cl)nc12 |r| Show InChI InChI=1S/C20H20Cl2N6O3/c1-23-18(31)20-6-11(20)13(14(29)15(20)30)28-8-25-12-16(26-19(22)27-17(12)28)24-7-9-3-2-4-10(21)5-9/h2-5,8,11,13-15,29-30H,6-7H2,1H3,(H,23,31)(H,24,26,27)/t11-,13-,14+,15+,20+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from human adenosine A3 receptor expressed in CHO cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50163020

((1S,2R,3S,4R,5S)-4-(6-(3-chlorobenzylamino)-2-chlo...)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Cl)c3)nc(Cl)nc12 |r| Show InChI InChI=1S/C20H20Cl2N6O3/c1-23-18(31)20-6-11(20)13(14(29)15(20)30)28-8-25-12-16(26-19(22)27-17(12)28)24-7-9-3-2-4-10(21)5-9/h2-5,8,11,13-15,29-30H,6-7H2,1H3,(H,23,31)(H,24,26,27)/t11-,13-,14+,15+,20+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18161

((1S,2S,7S,10R,11S,14S,15S)-14-hydroxy-2,15-dimethy...)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])CC(=O)CC[C@]12C |r| Show InChI InChI=1S/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12,14-17,21H,3-11H2,1-2H3/t12-,14-,15-,16-,17-,18-,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | -53.8 | n/a | n/a | 2.80 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Adenosine receptor A3
(Rattus norvegicus) | BDBM21221

((2S,3S,4R,5R)-5-(2-chloro-6-{[(3-iodophenyl)methyl...)Show SMILES CNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(NCc3cccc(I)c3)nc(Cl)nc12 Show InChI InChI=1S/C18H18ClIN6O4/c1-21-16(29)13-11(27)12(28)17(30-13)26-7-23-10-14(24-18(19)25-15(10)26)22-6-8-3-2-4-9(20)5-8/h2-5,7,11-13,17,27-28H,6H2,1H3,(H,21,29)(H,22,24,25)/t11-,12+,13-,17+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from rat A3 adenosine receptor |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218280

(5-chloro-N-((3R,4S)-1-(2-methoxyacetyl)-4-(4-(2-ox...)Show SMILES COCC(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C24H23ClN4O5S/c1-34-14-22(31)28-12-17(18(13-28)27-24(33)19-9-10-20(25)35-19)26-23(32)15-5-7-16(8-6-15)29-11-3-2-4-21(29)30/h2-11,17-18H,12-14H2,1H3,(H,26,32)(H,27,33)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50163025

((1S,2R,3S,4R,5S)-4-(6-(3-bromobenzylamino)-2-chlor...)Show SMILES CNC(=O)[C@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Br)c3)nc(Cl)nc12 Show InChI InChI=1S/C20H20BrClN6O3/c1-23-18(31)20-6-11(20)13(14(29)15(20)30)28-8-25-12-16(26-19(22)27-17(12)28)24-7-9-3-2-4-10(21)5-9/h2-5,8,11,13-15,29-30H,6-7H2,1H3,(H,23,31)(H,24,26,27)/t11-,13-,14+,15+,20-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from human adenosine A3 receptor expressed in CHO cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218315

((3R,4S)-methyl 3-(2-chlorothiophene-5-carboxamido)...)Show SMILES COC(=O)C1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O |w:4.3| Show InChI InChI=1S/C24H22ClN3O5S/c1-33-24(32)15-12-17(18(13-15)27-23(31)19-9-10-20(25)34-19)26-22(30)14-5-7-16(8-6-14)28-11-3-2-4-21(28)29/h2-11,15,17-18H,12-13H2,1H3,(H,26,30)(H,27,31)/t15?,17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216554
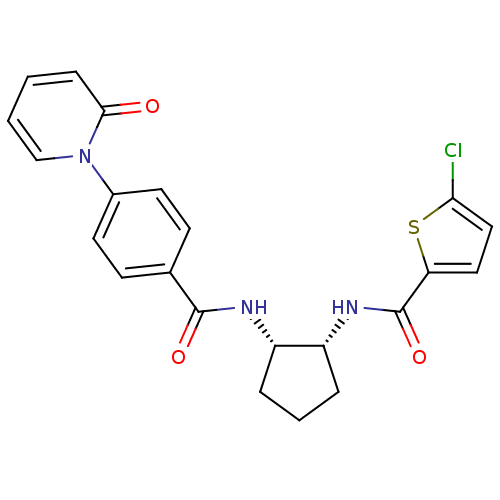
(5-CHLORO-N-((1R,2S)-2-(4-(2-OXOPYRIDIN-1(2H)-YL)BE...)Show SMILES Clc1ccc(s1)C(=O)N[C@@H]1CCC[C@@H]1NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C22H20ClN3O3S/c23-19-12-11-18(30-19)22(29)25-17-5-3-4-16(17)24-21(28)14-7-9-15(10-8-14)26-13-2-1-6-20(26)27/h1-2,6-13,16-17H,3-5H2,(H,24,28)(H,25,29)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216554

(5-CHLORO-N-((1R,2S)-2-(4-(2-OXOPYRIDIN-1(2H)-YL)BE...)Show SMILES Clc1ccc(s1)C(=O)N[C@@H]1CCC[C@@H]1NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C22H20ClN3O3S/c23-19-12-11-18(30-19)22(29)25-17-5-3-4-16(17)24-21(28)14-7-9-15(10-8-14)26-13-2-1-6-20(26)27/h1-2,6-13,16-17H,3-5H2,(H,24,28)(H,25,29)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216584
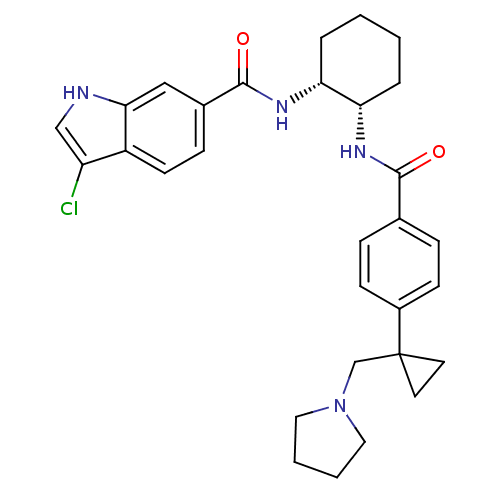
(3-chloro-N-((1R,2S)-2-(4-(1-(pyrrolidin-1-ylmethyl...)Show SMILES Clc1c[nH]c2cc(ccc12)C(=O)N[C@@H]1CCCC[C@@H]1NC(=O)c1ccc(cc1)C1(CN2CCCC2)CC1 Show InChI InChI=1S/C30H35ClN4O2/c31-24-18-32-27-17-21(9-12-23(24)27)29(37)34-26-6-2-1-5-25(26)33-28(36)20-7-10-22(11-8-20)30(13-14-30)19-35-15-3-4-16-35/h7-12,17-18,25-26,32H,1-6,13-16,19H2,(H,33,36)(H,34,37)/t25-,26+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218308
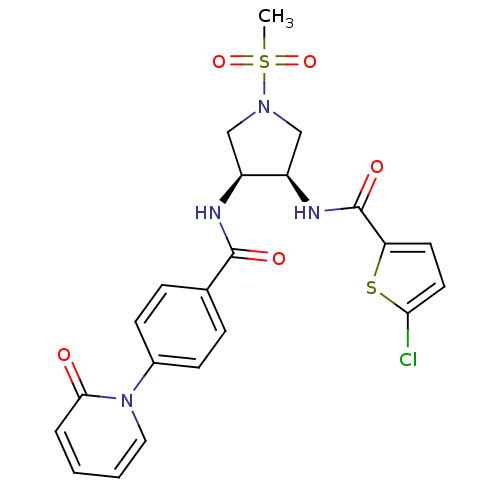
(5-chloro-N-((3R,4S)-1-(methylsulfonyl)-4-(4-(2-oxo...)Show SMILES CS(=O)(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C22H21ClN4O5S2/c1-34(31,32)26-12-16(17(13-26)25-22(30)18-9-10-19(23)33-18)24-21(29)14-5-7-15(8-6-14)27-11-3-2-4-20(27)28/h2-11,16-17H,12-13H2,1H3,(H,24,29)(H,25,30)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218282
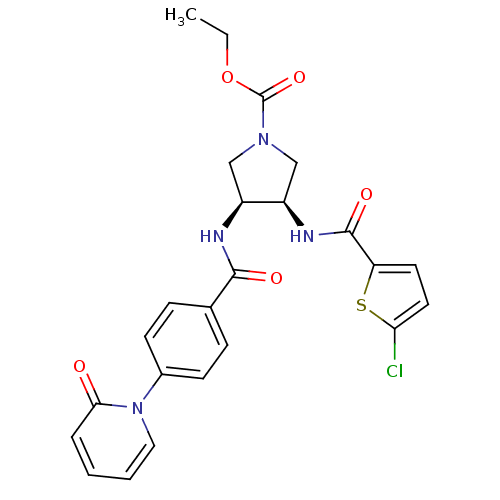
((3R,4S)-ethyl 3-(2-chlorothiophene-5-carboxamido)-...)Show SMILES CCOC(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C24H23ClN4O5S/c1-2-34-24(33)28-13-17(18(14-28)27-23(32)19-10-11-20(25)35-19)26-22(31)15-6-8-16(9-7-15)29-12-4-3-5-21(29)30/h3-12,17-18H,2,13-14H2,1H3,(H,26,31)(H,27,32)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216572
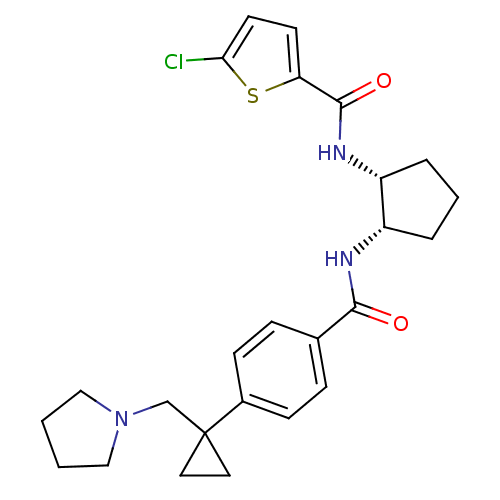
(5-chloro-N-((1R,2S)-2-(4-(1-(pyrrolidin-1-ylmethyl...)Show SMILES Clc1ccc(s1)C(=O)N[C@@H]1CCC[C@@H]1NC(=O)c1ccc(cc1)C1(CN2CCCC2)CC1 Show InChI InChI=1S/C25H30ClN3O2S/c26-22-11-10-21(32-22)24(31)28-20-5-3-4-19(20)27-23(30)17-6-8-18(9-7-17)25(12-13-25)16-29-14-1-2-15-29/h6-11,19-20H,1-5,12-16H2,(H,27,30)(H,28,31)/t19-,20+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50389134
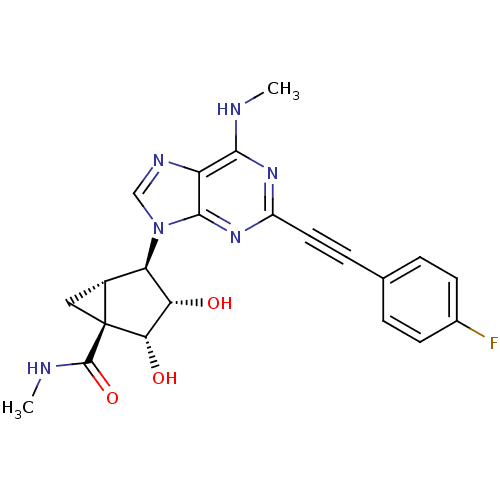
(CHEMBL2064639)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NC)nc(nc12)C#Cc1ccc(F)cc1 |r| Show InChI InChI=1S/C22H21FN6O3/c1-24-19-15-20(28-14(27-19)8-5-11-3-6-12(23)7-4-11)29(10-26-15)16-13-9-22(13,21(32)25-2)18(31)17(16)30/h3-4,6-7,10,13,16-18,30-31H,9H2,1-2H3,(H,25,32)(H,24,27,28)/t13-,16-,17+,18+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218312
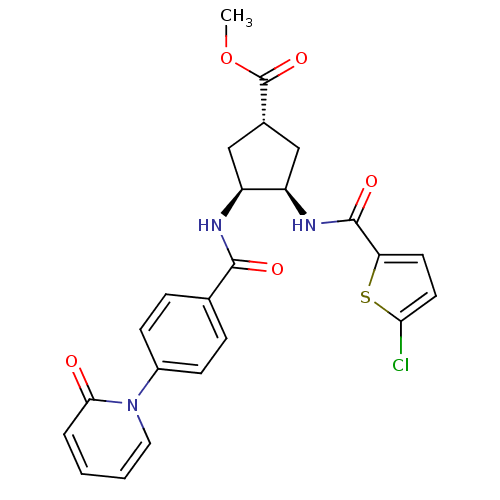
((1S,3R,4S)-methyl 3-(2-chlorothiophene-5-carboxami...)Show SMILES COC(=O)[C@@H]1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C24H22ClN3O5S/c1-33-24(32)15-12-17(18(13-15)27-23(31)19-9-10-20(25)34-19)26-22(30)14-5-7-16(8-6-14)28-11-3-2-4-21(28)29/h2-11,15,17-18H,12-13H2,1H3,(H,26,30)(H,27,31)/t15-,17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218290
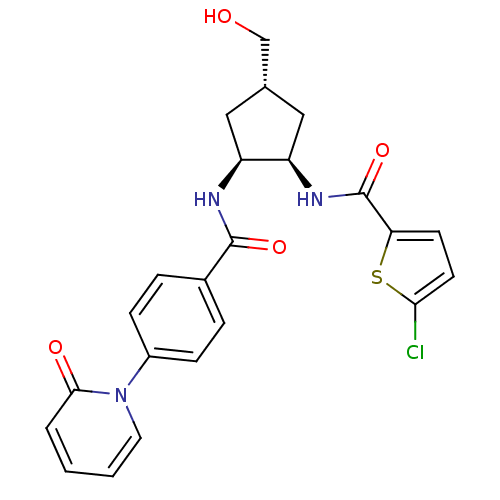
(5-chloro-N-((1R,2S,4S)-4-(hydroxymethyl)-2-(4-(2-o...)Show SMILES OC[C@@H]1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C23H22ClN3O4S/c24-20-9-8-19(32-20)23(31)26-18-12-14(13-28)11-17(18)25-22(30)15-4-6-16(7-5-15)27-10-2-1-3-21(27)29/h1-10,14,17-18,28H,11-13H2,(H,25,30)(H,26,31)/t14-,17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218300
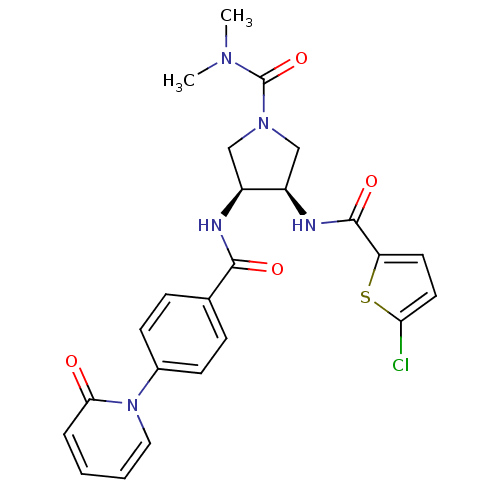
((3R,4S)-3-(2-chlorothiophene-5-carboxamido)-N,N-di...)Show SMILES CN(C)C(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C24H24ClN5O4S/c1-28(2)24(34)29-13-17(18(14-29)27-23(33)19-10-11-20(25)35-19)26-22(32)15-6-8-16(9-7-15)30-12-4-3-5-21(30)31/h3-12,17-18H,13-14H2,1-2H3,(H,26,32)(H,27,33)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50389135

(CHEMBL2064640)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NC)nc(nc12)C#Cc1ccccc1Cl |r| Show InChI InChI=1S/C22H21ClN6O3/c1-24-19-15-20(28-14(27-19)8-7-11-5-3-4-6-13(11)23)29(10-26-15)16-12-9-22(12,21(32)25-2)18(31)17(16)30/h3-6,10,12,16-18,30-31H,9H2,1-2H3,(H,25,32)(H,24,27,28)/t12-,16-,17+,18+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218291
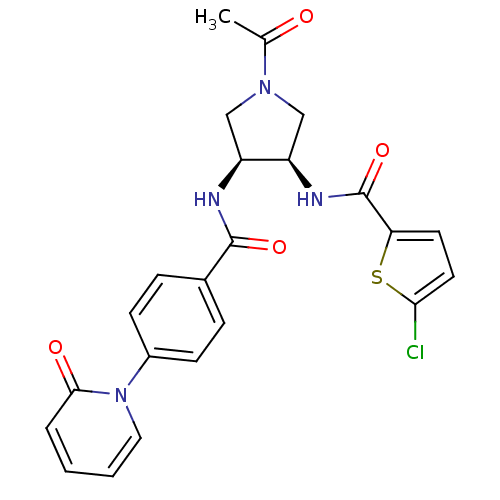
(CHEMBL394129 | N-((3R,4S)-1-acetyl-4-(4-(2-oxopyri...)Show SMILES CC(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C23H21ClN4O4S/c1-14(29)27-12-17(18(13-27)26-23(32)19-9-10-20(24)33-19)25-22(31)15-5-7-16(8-6-15)28-11-3-2-4-21(28)30/h2-11,17-18H,12-13H2,1H3,(H,25,31)(H,26,32)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218283
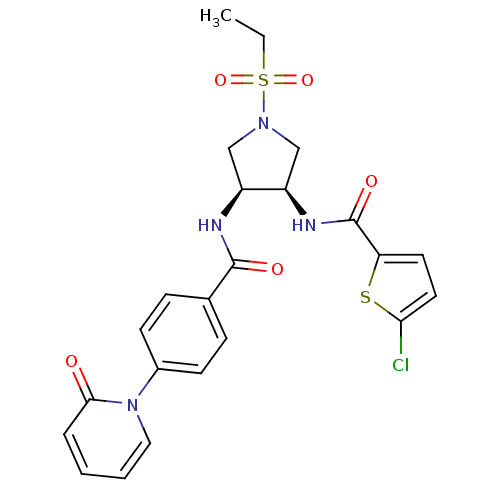
(5-chloro-N-((3R,4S)-1-(ethylsulfonyl)-4-(4-(2-oxop...)Show SMILES CCS(=O)(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C23H23ClN4O5S2/c1-2-35(32,33)27-13-17(18(14-27)26-23(31)19-10-11-20(24)34-19)25-22(30)15-6-8-16(9-7-15)28-12-4-3-5-21(28)29/h3-12,17-18H,2,13-14H2,1H3,(H,25,30)(H,26,31)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216556
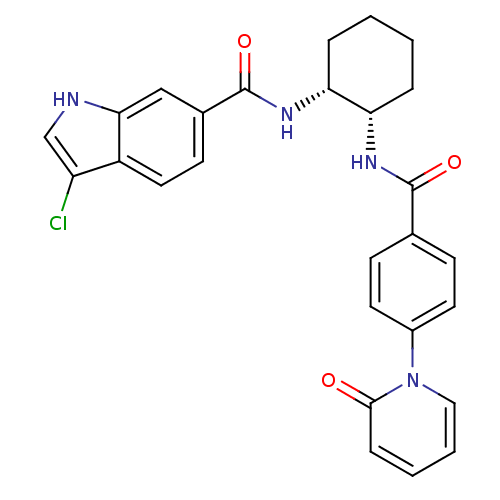
(3-CHLORO-N-((1R,2S) -2-(4-(2-OXOPYRIDIN-1(2H)-YL)B...)Show SMILES Clc1c[nH]c2cc(ccc12)C(=O)N[C@@H]1CCCC[C@@H]1NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C27H25ClN4O3/c28-21-16-29-24-15-18(10-13-20(21)24)27(35)31-23-6-2-1-5-22(23)30-26(34)17-8-11-19(12-9-17)32-14-4-3-7-25(32)33/h3-4,7-16,22-23,29H,1-2,5-6H2,(H,30,34)(H,31,35)/t22-,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50163031
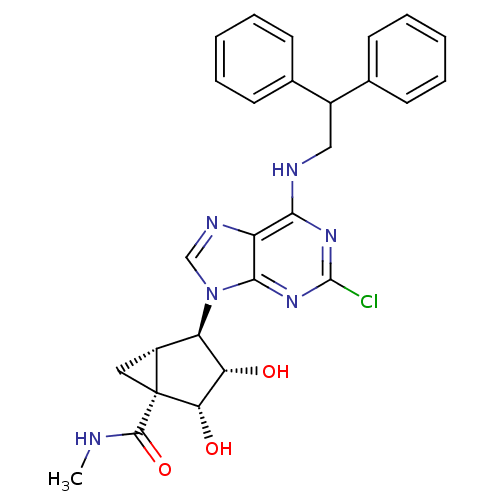
((1S,2R,3S,4R,5S)-4-(2-chloro-6-(2,2-diphenylethyla...)Show SMILES CNC(=O)[C@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCC(c3ccccc3)c3ccccc3)nc(Cl)nc12 Show InChI InChI=1S/C27H27ClN6O3/c1-29-25(37)27-12-18(27)20(21(35)22(27)36)34-14-31-19-23(32-26(28)33-24(19)34)30-13-17(15-8-4-2-5-9-15)16-10-6-3-7-11-16/h2-11,14,17-18,20-22,35-36H,12-13H2,1H3,(H,29,37)(H,30,32,33)/t18-,20-,21+,22+,27-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from human adenosine A3 receptor expressed in CHO cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216604

(3-chloro-N-((1R,2S)-2-(4-(2-oxopyridin-1(2H)-yl)be...)Show SMILES Clc1c[nH]c2cc(ccc12)C(=O)N[C@@H]1CCC[C@@H]1NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C26H23ClN4O3/c27-20-15-28-23-14-17(9-12-19(20)23)26(34)30-22-5-3-4-21(22)29-25(33)16-7-10-18(11-8-16)31-13-2-1-6-24(31)32/h1-2,6-15,21-22,28H,3-5H2,(H,29,33)(H,30,34)/t21-,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216579
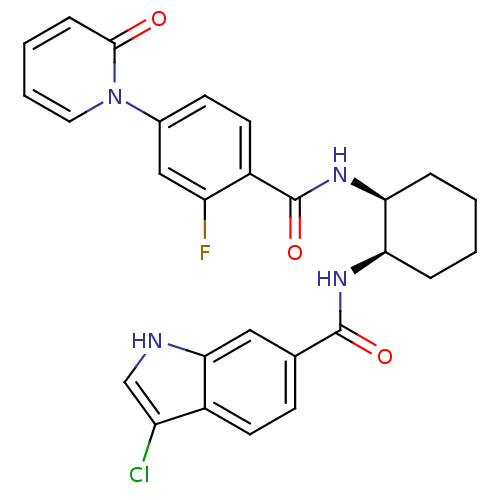
(CHEMBL401024 | cis-3-chloro-N-(2-(2-fluoro-4-(2-ox...)Show SMILES Fc1cc(ccc1C(=O)N[C@H]1CCCC[C@H]1NC(=O)c1ccc2c(Cl)c[nH]c2c1)-n1ccccc1=O Show InChI InChI=1S/C27H24ClFN4O3/c28-20-15-30-24-13-16(8-10-18(20)24)26(35)31-22-5-1-2-6-23(22)32-27(36)19-11-9-17(14-21(19)29)33-12-4-3-7-25(33)34/h3-4,7-15,22-23,30H,1-2,5-6H2,(H,31,35)(H,32,36)/t22-,23+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218282

((3R,4S)-ethyl 3-(2-chlorothiophene-5-carboxamido)-...)Show SMILES CCOC(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C24H23ClN4O5S/c1-2-34-24(33)28-13-17(18(14-28)27-23(32)19-10-11-20(25)35-19)26-22(31)15-6-8-16(9-7-15)29-12-4-3-5-21(29)30/h3-12,17-18H,2,13-14H2,1H3,(H,26,31)(H,27,32)/t17-,18+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a in presence of tripeptide substrate at 25 degC |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218306
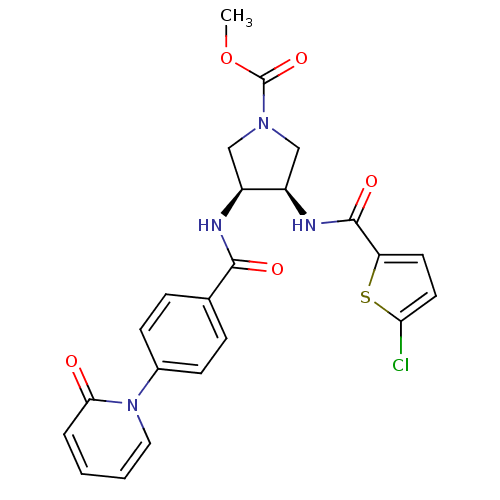
((3R,4S)-methyl 3-(2-chlorothiophene-5-carboxamido)...)Show SMILES COC(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C23H21ClN4O5S/c1-33-23(32)27-12-16(17(13-27)26-22(31)18-9-10-19(24)34-18)25-21(30)14-5-7-15(8-6-14)28-11-3-2-4-20(28)29/h2-11,16-17H,12-13H2,1H3,(H,25,30)(H,26,31)/t16-,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Rattus norvegicus) | BDBM50163025

((1S,2R,3S,4R,5S)-4-(6-(3-bromobenzylamino)-2-chlor...)Show SMILES CNC(=O)[C@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Br)c3)nc(Cl)nc12 Show InChI InChI=1S/C20H20BrClN6O3/c1-23-18(31)20-6-11(20)13(14(29)15(20)30)28-8-25-12-16(26-19(22)27-17(12)28)24-7-9-3-2-4-10(21)5-9/h2-5,8,11,13-15,29-30H,6-7H2,1H3,(H,23,31)(H,24,26,27)/t11-,13-,14+,15+,20-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.760 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from rat A3 adenosine receptor |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50163004

((1S,2R,3S,4R,5S)-4-(2-chloro-6-(2-phenylcyclopropy...)Show SMILES CNC(=O)[C@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NC3CC3c3ccccc3)nc(Cl)nc12 Show InChI InChI=1S/C22H23ClN6O3/c1-24-20(32)22-8-12(22)15(16(30)17(22)31)29-9-25-14-18(27-21(23)28-19(14)29)26-13-7-11(13)10-5-3-2-4-6-10/h2-6,9,11-13,15-17,30-31H,7-8H2,1H3,(H,24,32)(H,26,27,28)/t11?,12-,13?,15-,16+,17+,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from human adenosine A3 receptor expressed in CHO cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Mus musculus) | BDBM50100418
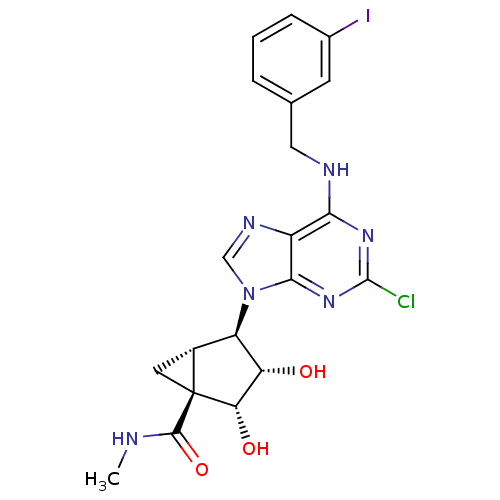
((1S,2R,3S,4R,5S)-4-(6-(3-iodobenzylamino)-2-chloro...)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(I)c3)nc(Cl)nc12 |r| Show InChI InChI=1S/C20H20ClIN6O3/c1-23-18(31)20-6-11(20)13(14(29)15(20)30)28-8-25-12-16(26-19(21)27-17(12)28)24-7-9-3-2-4-10(22)5-9/h2-5,8,11,13-15,29-30H,6-7H2,1H3,(H,23,31)(H,24,26,27)/t11-,13-,14+,15+,20+/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [125I]N6-(-amino-3-iodobenzyl)adenosine-5'-N-methyl-uronamide from mouse recombinant adenosine A3 receptor expressed in HEK293 cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Mus musculus) | BDBM50389126

(CHEMBL2064658)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Cl)c3)nc(nc12)C#Cc1ccc(N)cc1 |r| Show InChI InChI=1S/C28H26ClN7O3/c1-31-27(39)28-12-19(28)22(23(37)24(28)38)36-14-33-21-25(32-13-16-3-2-4-17(29)11-16)34-20(35-26(21)36)10-7-15-5-8-18(30)9-6-15/h2-6,8-9,11,14,19,22-24,37-38H,12-13,30H2,1H3,(H,31,39)(H,32,34,35)/t19-,22-,23+,24+,28+/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]CGS21680 from mouse adenosine A3 receptor expressed in HEK293 cells after 60 mins by liquid scintillation counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50377678
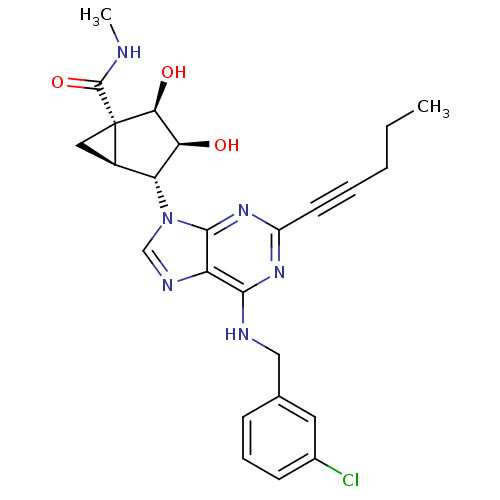
(CHEMBL258421)Show SMILES CCCC#Cc1nc(NCc2cccc(Cl)c2)c2ncn([C@@H]3[C@H]4C[C@]4([C@@H](O)[C@H]3O)C(=O)NC)c2n1 Show InChI InChI=1S/C25H27ClN6O3/c1-3-4-5-9-17-30-22(28-12-14-7-6-8-15(26)10-14)18-23(31-17)32(13-29-18)19-16-11-25(16,24(35)27-2)21(34)20(19)33/h6-8,10,13,16,19-21,33-34H,3-4,11-12H2,1-2H3,(H,27,35)(H,28,30,31)/t16-,19-,20+,21+,25+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from human adenosine A3 receptor expressed in CHO cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218319
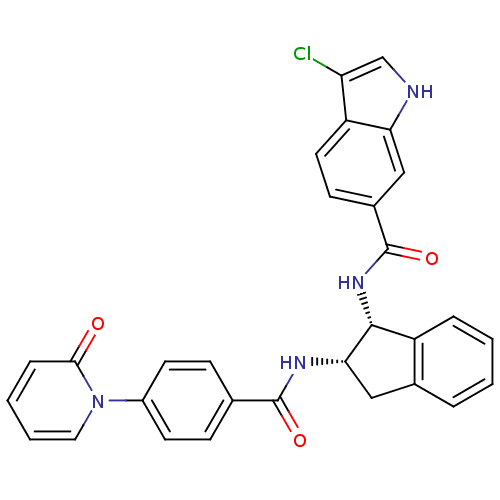
(3-chloro-N-((1R,2S)-2-(4-(2-oxopyridin-1(2H)-yl)be...)Show SMILES Clc1c[nH]c2cc(ccc12)C(=O)N[C@H]1[C@H](Cc2ccccc12)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C30H23ClN4O3/c31-24-17-32-25-16-20(10-13-23(24)25)30(38)34-28-22-6-2-1-5-19(22)15-26(28)33-29(37)18-8-11-21(12-9-18)35-14-4-3-7-27(35)36/h1-14,16-17,26,28,32H,15H2,(H,33,37)(H,34,38)/t26-,28+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.840 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50389130

(CHEMBL2064635)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NC)nc(nc12)C#Cc1ccccc1 |r| Show InChI InChI=1S/C22H22N6O3/c1-23-19-15-20(27-14(26-19)9-8-12-6-4-3-5-7-12)28(11-25-15)16-13-10-22(13,21(31)24-2)18(30)17(16)29/h3-7,11,13,16-18,29-30H,10H2,1-2H3,(H,24,31)(H,23,26,27)/t13-,16-,17+,18+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Mus musculus) | BDBM50377679
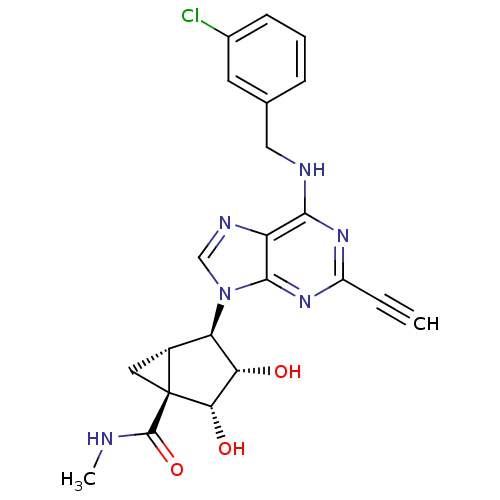
(CHEMBL402574)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Cl)c3)nc(nc12)C#C Show InChI InChI=1S/C22H21ClN6O3/c1-3-14-27-19(25-9-11-5-4-6-12(23)7-11)15-20(28-14)29(10-26-15)16-13-8-22(13,21(32)24-2)18(31)17(16)30/h1,4-7,10,13,16-18,30-31H,8-9H2,2H3,(H,24,32)(H,25,27,28)/t13-,16-,17+,18+,22+/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [125I]N6-(-amino-3-iodobenzyl)adenosine-5'-N-methyl-uronamide from mouse recombinant adenosine A3 receptor expressed in HEK293 cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Rattus norvegicus) | BDBM50163014

((1S,2R,3S,4R,5S)-4-(6-(2,5-dimethoxybenzylamino)-2...)Show SMILES CNC(=O)[C@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cc(OC)ccc3OC)nc(Cl)nc12 Show InChI InChI=1S/C22H25ClN6O5/c1-24-20(32)22-7-12(22)15(16(30)17(22)31)29-9-26-14-18(27-21(23)28-19(14)29)25-8-10-6-11(33-2)4-5-13(10)34-3/h4-6,9,12,15-17,30-31H,7-8H2,1-3H3,(H,24,32)(H,25,27,28)/t12-,15-,16+,17+,22-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from rat A3 adenosine receptor |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Mus musculus) | BDBM50163025

((1S,2R,3S,4R,5S)-4-(6-(3-bromobenzylamino)-2-chlor...)Show SMILES CNC(=O)[C@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Br)c3)nc(Cl)nc12 Show InChI InChI=1S/C20H20BrClN6O3/c1-23-18(31)20-6-11(20)13(14(29)15(20)30)28-8-25-12-16(26-19(22)27-17(12)28)24-7-9-3-2-4-10(21)5-9/h2-5,8,11,13-15,29-30H,6-7H2,1H3,(H,23,31)(H,24,26,27)/t11-,13-,14+,15+,20-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [125I]N6-(-amino-3-iodobenzyl)adenosine-5'-N-methyl-uronamide from mouse recombinant adenosine A3 receptor expressed in HEK293 cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50389137
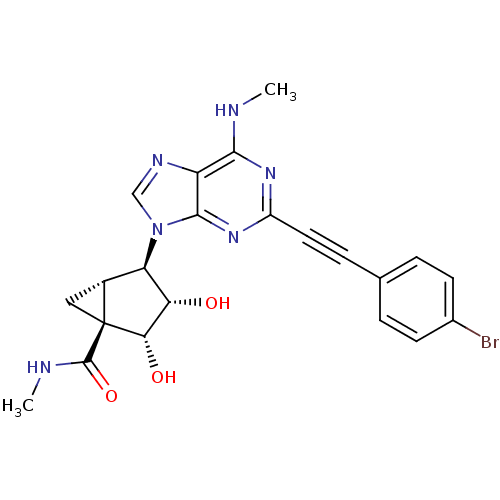
(CHEMBL2064643)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NC)nc(nc12)C#Cc1ccc(Br)cc1 |r| Show InChI InChI=1S/C22H21BrN6O3/c1-24-19-15-20(28-14(27-19)8-5-11-3-6-12(23)7-4-11)29(10-26-15)16-13-9-22(13,21(32)25-2)18(31)17(16)30/h3-4,6-7,10,13,16-18,30-31H,9H2,1-2H3,(H,25,32)(H,24,27,28)/t13-,16-,17+,18+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50218294

((3R,4S)-2-methoxyethyl 3-(2-chlorothiophene-5-carb...)Show SMILES COCCOC(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O Show InChI InChI=1S/C25H25ClN4O6S/c1-35-12-13-36-25(34)29-14-18(19(15-29)28-24(33)20-9-10-21(26)37-20)27-23(32)16-5-7-17(8-6-16)30-11-3-2-4-22(30)31/h2-11,18-19H,12-15H2,1H3,(H,27,32)(H,28,33)/t18-,19+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 5041-8 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.020
BindingDB Entry DOI: 10.7270/Q23J3CPQ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216583
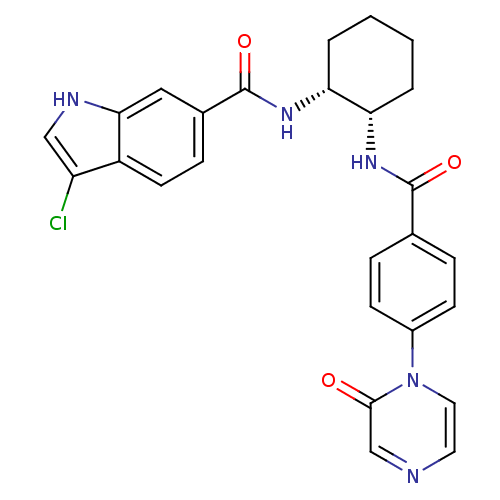
(3-chloro-N-((1R,2S)-2-(4-(2-oxopyrazin-1(2H)-yl)be...)Show SMILES Clc1c[nH]c2cc(ccc12)C(=O)N[C@@H]1CCCC[C@@H]1NC(=O)c1ccc(cc1)-n1ccncc1=O Show InChI InChI=1S/C26H24ClN5O3/c27-20-14-29-23-13-17(7-10-19(20)23)26(35)31-22-4-2-1-3-21(22)30-25(34)16-5-8-18(9-6-16)32-12-11-28-15-24(32)33/h5-15,21-22,29H,1-4H2,(H,30,34)(H,31,35)/t21-,22+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50216620
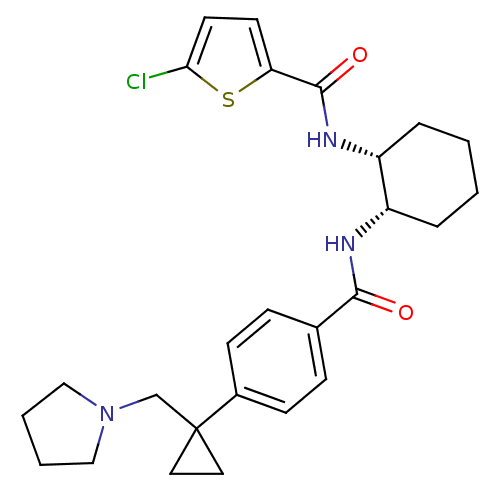
(5-chloro-N-((1R,2S)-2-(4-(1-(pyrrolidin-1-ylmethyl...)Show SMILES Clc1ccc(s1)C(=O)N[C@@H]1CCCC[C@@H]1NC(=O)c1ccc(cc1)C1(CN2CCCC2)CC1 Show InChI InChI=1S/C26H32ClN3O2S/c27-23-12-11-22(33-23)25(32)29-21-6-2-1-5-20(21)28-24(31)18-7-9-19(10-8-18)26(13-14-26)17-30-15-3-4-16-30/h7-12,20-21H,1-6,13-17H2,(H,28,31)(H,29,32)/t20-,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Company
Curated by ChEMBL
| Assay Description
Inhibition of human factor 10a |
Bioorg Med Chem Lett 17: 4419-27 (2007)
Article DOI: 10.1016/j.bmcl.2007.06.029
BindingDB Entry DOI: 10.7270/Q2416XVV |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50389133
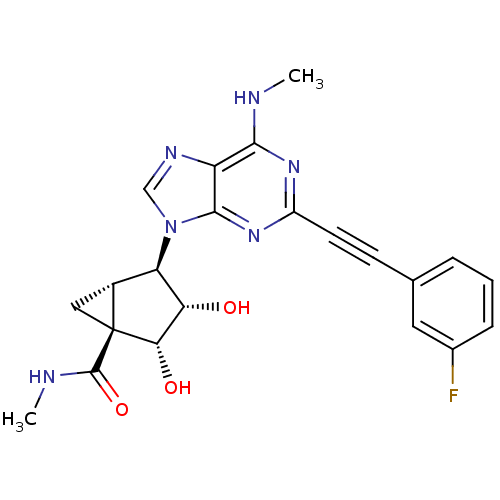
(CHEMBL2064638)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NC)nc(nc12)C#Cc1cccc(F)c1 |r| Show InChI InChI=1S/C22H21FN6O3/c1-24-19-15-20(28-14(27-19)7-6-11-4-3-5-12(23)8-11)29(10-26-15)16-13-9-22(13,21(32)25-2)18(31)17(16)30/h3-5,8,10,13,16-18,30-31H,9H2,1-2H3,(H,25,32)(H,24,27,28)/t13-,16-,17+,18+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50389132

(CHEMBL2064637)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NC)nc(nc12)C#Cc1ccccc1F |r| Show InChI InChI=1S/C22H21FN6O3/c1-24-19-15-20(28-14(27-19)8-7-11-5-3-4-6-13(11)23)29(10-26-15)16-12-9-22(12,21(32)25-2)18(31)17(16)30/h3-6,10,12,16-18,30-31H,9H2,1-2H3,(H,25,32)(H,24,27,28)/t12-,16-,17+,18+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of [3H]I-AB-MECA from human recombinant adenosine A3 receptor expressed in CHO cells after 60 mins by gamma counting |
J Med Chem 55: 4847-60 (2012)
Article DOI: 10.1021/jm300396n
BindingDB Entry DOI: 10.7270/Q2765GCG |
More data for this
Ligand-Target Pair | |
Adenosine receptor A3
(Homo sapiens (Human)) | BDBM50377684
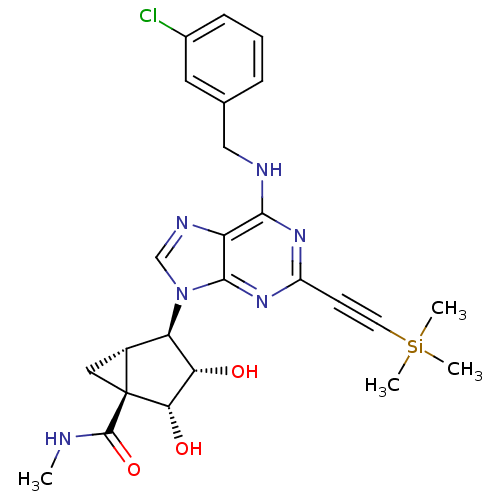
(CHEMBL257405)Show SMILES CNC(=O)[C@@]12C[C@@H]1[C@H]([C@H](O)[C@@H]2O)n1cnc2c(NCc3cccc(Cl)c3)nc(nc12)C#C[Si](C)(C)C Show InChI InChI=1S/C25H29ClN6O3Si/c1-27-24(35)25-11-16(25)19(20(33)21(25)34)32-13-29-18-22(28-12-14-6-5-7-15(26)10-14)30-17(31-23(18)32)8-9-36(2,3)4/h5-7,10,13,16,19-21,33-34H,11-12H2,1-4H3,(H,27,35)(H,28,30,31)/t16-,19-,20+,21+,25+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.980 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Displacement of radioligand from human adenosine A3 receptor expressed in CHO cells |
Bioorg Med Chem Lett 18: 2813-9 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.001
BindingDB Entry DOI: 10.7270/Q2XK8GFH |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18162
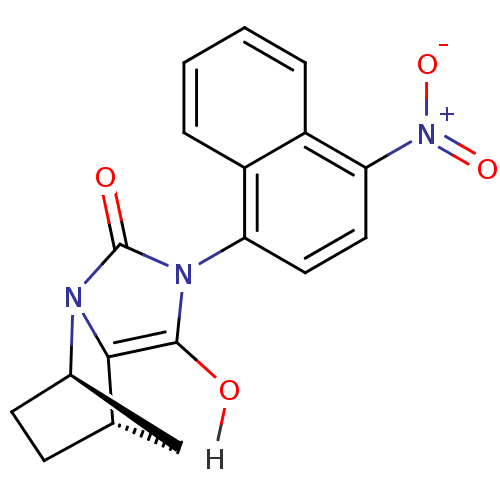
((1R,6R,7S)-4-(4-nitronaphthalen-1-yl)-2,4-diazatri...)Show SMILES Oc1c2[C@H]3CC[C@H](C3)n2c(=O)n1-c1ccc([N+]([O-])=O)c2ccccc12 |r,wU:6.6,3.7,(12.2,-7.28,;12.6,-8.77,;11.56,-9.96,;10.02,-9.96,;9.25,-11.29,;10.02,-12.62,;11.56,-12.62,;10.02,-11.73,;12.33,-11.29,;13.87,-10.92,;15.03,-11.93,;14,-9.4,;15.49,-9,;16.07,-7.57,;17.59,-7.36,;18.54,-8.57,;20.06,-8.36,;20.83,-9.69,;20.64,-6.93,;17.96,-10,;18.91,-11.21,;18.34,-12.64,;16.81,-12.86,;15.86,-11.64,;16.44,-10.22,)| Show InChI InChI=1S/C18H15N3O4/c22-17-16-10-5-6-11(9-10)19(16)18(23)20(17)14-7-8-15(21(24)25)13-4-2-1-3-12(13)14/h1-4,7-8,10-11,22H,5-6,9H2/t10-,11+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | -50.9 | n/a | n/a | 385 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data