| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM8963 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_552460 (CHEMBL1002720) |
|---|
| Ki | 1.30±n/a nM |
|---|
| Citation |  Butini, S; Campiani, G; Borriello, M; Gemma, S; Panico, A; Persico, M; Catalanotti, B; Ros, S; Brindisi, M; Agnusdei, M; Fiorini, I; Nacci, V; Novellino, E; Belinskaya, T; Saxena, A; Fattorusso, C Exploiting protein fluctuations at the active-site gorge of human cholinesterases: further optimization of the design strategy to develop extremely potent inhibitors. J Med Chem51:3154-70 (2008) [PubMed] Article Butini, S; Campiani, G; Borriello, M; Gemma, S; Panico, A; Persico, M; Catalanotti, B; Ros, S; Brindisi, M; Agnusdei, M; Fiorini, I; Nacci, V; Novellino, E; Belinskaya, T; Saxena, A; Fattorusso, C Exploiting protein fluctuations at the active-site gorge of human cholinesterases: further optimization of the design strategy to develop extremely potent inhibitors. J Med Chem51:3154-70 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_BOVIN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase precursor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67659.62 |
|---|
| Organism: | Bos taurus (bovine) |
|---|
| Description: | n/a |
|---|
| Residue: | 613 |
|---|
| Sequence: | MRPPWCPLHTPSLTPPLLLLLFLIGGGAEAEGPEDPELLVMVRGGRLRGLRLMAPRGPVS
AFLGIPFAEPPVGPRRFLPPEPKRPWPGVLNATAFQSVCYQYVDTLYPGFEGTEMWNPNR
ELSEDCLYLNVWTPYPRPSSPTPVLVWIYGGGFYSGASSLDVYDGRFLTQAEGTVLVSMN
YRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASVG
MHLLSPPSRGLFHRAVLQSGAPNGPWATVGVGEARRRATLLARLVGCPPGGAGGNDTELV
ACLRARPAQDLVDHEWRVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVGV
VKDEGSYFLVYGAPGFSKDNESLISRAQFLAGVRVGVPQASDLAAEAVVLHYTDWLHPED
PARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYIFEHRASTLSWPLWMGVPHGYE
IEFIFGLPLEPSLNYTIEERTFAQRLMRYWANFARTGDPNDPRDPKAPQWPPYTAGAQQY
VSLNLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKNQ
FDHYSKQDRCSDL
|
|
|
|---|
| BDBM8963 |
|---|
| n/a |
|---|
| Name | BDBM8963 |
|---|
| Synonyms: | CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7-(1,2,3,4-tetrahydroacridin-9-ylamino)heptyl]-1,2,3,4-tetrahydroacridin-9-amine | Tacrine Dimer 3b | Tacrine-Based Inhibitor 2f | tacrine homobivalent compound 3a |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H40N4 |
|---|
| Mol. Mass. | 492.6975 |
|---|
| SMILES | C(CCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 |
|---|
| Structure | 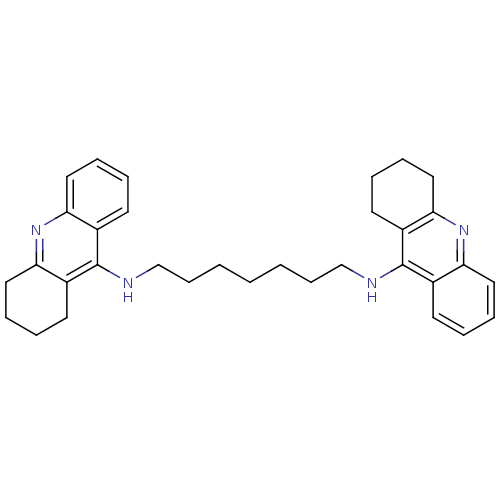
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Butini, S; Campiani, G; Borriello, M; Gemma, S; Panico, A; Persico, M; Catalanotti, B; Ros, S; Brindisi, M; Agnusdei, M; Fiorini, I; Nacci, V; Novellino, E; Belinskaya, T; Saxena, A; Fattorusso, C Exploiting protein fluctuations at the active-site gorge of human cholinesterases: further optimization of the design strategy to develop extremely potent inhibitors. J Med Chem51:3154-70 (2008) [PubMed] Article
Butini, S; Campiani, G; Borriello, M; Gemma, S; Panico, A; Persico, M; Catalanotti, B; Ros, S; Brindisi, M; Agnusdei, M; Fiorini, I; Nacci, V; Novellino, E; Belinskaya, T; Saxena, A; Fattorusso, C Exploiting protein fluctuations at the active-site gorge of human cholinesterases: further optimization of the design strategy to develop extremely potent inhibitors. J Med Chem51:3154-70 (2008) [PubMed] Article