| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sodium-dependent dopamine transporter |
|---|
| Ligand | BDBM50243140 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_492350 (CHEMBL948346) |
|---|
| IC50 | 17.5±n/a nM |
|---|
| Citation |  Jin, C; Navarro, HA; Page, K; Carroll, FI Synthesis and monoamine transporter binding properties of 2beta-[3'-(substituted benzyl)isoxazol-5-yl]- and 2beta-[3'-methyl-4'-(substituted phenyl)isoxazol-5-yl]-3beta-(substituted phenyl)tropanes. Bioorg Med Chem16:6682-8 (2008) [PubMed] Article Jin, C; Navarro, HA; Page, K; Carroll, FI Synthesis and monoamine transporter binding properties of 2beta-[3'-(substituted benzyl)isoxazol-5-yl]- and 2beta-[3'-methyl-4'-(substituted phenyl)isoxazol-5-yl]-3beta-(substituted phenyl)tropanes. Bioorg Med Chem16:6682-8 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sodium-dependent dopamine transporter |
|---|
| Name: | Sodium-dependent dopamine transporter |
|---|
| Synonyms: | DA transporter | DAT | DAT1 | Dopamine transporter (DAT) | Dopamine transporter protein (DAT) | Monoamine transporter | SC6A3_HUMAN | SLC6A3 | Sodium-dependent dopamine transporter | Sodium-dependent dopamine transporter (DAT) | Solute carrier family 6 member 3 |
|---|
| Type: | Multi-pass membrane protein |
|---|
| Mol. Mass.: | 68497.11 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q01959 |
|---|
| Residue: | 620 |
|---|
| Sequence: | MSKSKCSVGLMSSVVAPAKEPNAVGPKEVELILVKEQNGVQLTSSTLTNPRQSPVEAQDR
ETWGKKIDFLLSVIGFAVDLANVWRFPYLCYKNGGGAFLVPYLLFMVIAGMPLFYMELAL
GQFNREGAAGVWKICPILKGVGFTVILISLYVGFFYNVIIAWALHYLFSSFTTELPWIHC
NNSWNSPNCSDAHPGDSSGDSSGLNDTFGTTPAAEYFERGVLHLHQSHGIDDLGPPRWQL
TACLVLVIVLLYFSLWKGVKTSGKVVWITATMPYVVLTALLLRGVTLPGAIDGIRAYLSV
DFYRLCEASVWIDAATQVCFSLGVGFGVLIAFSSYNKFTNNCYRDAIVTTSINSLTSFSS
GFVVFSFLGYMAQKHSVPIGDVAKDGPGLIFIIYPEAIATLPLSSAWAVVFFIMLLTLGI
DSAMGGMESVITGLIDEFQLLHRHRELFTLFIVLATFLLSLFCVTNGGIYVFTLLDHFAA
GTSILFGVLIEAIGVAWFYGVGQFSDDIQQMTGQRPSLYWRLCWKLVSPCFLLFVVVVSI
VTFRPPHYGAYIFPDWANALGWVIATSSMAMVPIYAAYKFCSLPGSFREKLAYAIAPEKD
RELVDRGEVRQFTLRHWLKV
|
|
|
|---|
| BDBM50243140 |
|---|
| n/a |
|---|
| Name | BDBM50243140 |
|---|
| Synonyms: | 3beta-(4-Chlorophenyl)-2beta-[3'-(4-chlorobenzyl)isoxazol-5-yl]-tropanehdrochloride | CHEMBL520035 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H24Cl2N2O |
|---|
| Mol. Mass. | 427.366 |
|---|
| SMILES | CN1C2CCC1[C@H]([C@H](C2)c1ccc(Cl)cc1)c1cc(Cc2ccc(Cl)cc2)no1 |r,TLB:16:6:1:4.3| |
|---|
| Structure | 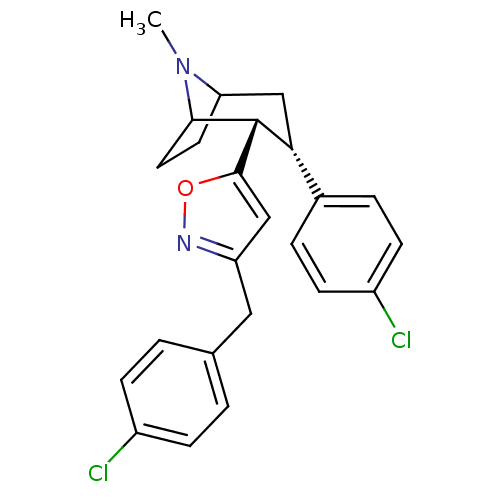
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jin, C; Navarro, HA; Page, K; Carroll, FI Synthesis and monoamine transporter binding properties of 2beta-[3'-(substituted benzyl)isoxazol-5-yl]- and 2beta-[3'-methyl-4'-(substituted phenyl)isoxazol-5-yl]-3beta-(substituted phenyl)tropanes. Bioorg Med Chem16:6682-8 (2008) [PubMed] Article
Jin, C; Navarro, HA; Page, K; Carroll, FI Synthesis and monoamine transporter binding properties of 2beta-[3'-(substituted benzyl)isoxazol-5-yl]- and 2beta-[3'-methyl-4'-(substituted phenyl)isoxazol-5-yl]-3beta-(substituted phenyl)tropanes. Bioorg Med Chem16:6682-8 (2008) [PubMed] Article