| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Vasopressin V1b receptor |
|---|
| Ligand | BDBM50299343 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_590267 (CHEMBL1052802) |
|---|
| Ki | 0.6±n/a nM |
|---|
| Citation |  Scott, JD; Miller, MW; Li, SW; Lin, SI; Vaccaro, HA; Hong, L; Mullins, DE; Guzzi, M; Weinstein, J; Hodgson, RA; Varty, GB; Stamford, AW; Chan, TY; McKittrick, BA; Greenlee, WJ; Priestley, T; Parker, EM Tetrahydroquinoline sulfonamides as vasopressin 1b receptor antagonists. Bioorg Med Chem Lett19:6018-22 (2009) [PubMed] Article Scott, JD; Miller, MW; Li, SW; Lin, SI; Vaccaro, HA; Hong, L; Mullins, DE; Guzzi, M; Weinstein, J; Hodgson, RA; Varty, GB; Stamford, AW; Chan, TY; McKittrick, BA; Greenlee, WJ; Priestley, T; Parker, EM Tetrahydroquinoline sulfonamides as vasopressin 1b receptor antagonists. Bioorg Med Chem Lett19:6018-22 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Vasopressin V1b receptor |
|---|
| Name: | Vasopressin V1b receptor |
|---|
| Synonyms: | AVPR V1b | AVPR V3 | AVPR1B | AVPR3 | Antidiuretic hormone receptor 1b | V1BR_HUMAN | V1bR | VASOPRESSIN V1B | VPR3 | Vasopressin V1b receptor | Vasopressin V1b receptor (V1b) | Vasopressin V3 | Vasopressin V3 receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 46985.01 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P47901 |
|---|
| Residue: | 424 |
|---|
| Sequence: | MDSGPLWDANPTPRGTLSAPNATTPWLGRDEELAKVEIGVLATVLVLATGGNLAVLLTLG
QLGRKRSRMHLFVLHLALTDLAVALFQVLPQLLWDITYRFQGPDLLCRAVKYLQVLSMFA
STYMLLAMTLDRYLAVCHPLRSLQQPGQSTYLLIAAPWLLAAIFSLPQVFIFSLREVIQG
SGVLDCWADFGFPWGPRAYLTWTTLAIFVLPVTMLTACYSLICHEICKNLKVKTQAWRVG
GGGWRTWDRPSPSTLAATTRGLPSRVSSINTISRAKIRTVKMTFVIVLAYIACWAPFFSV
QMWSVWDKNAPDEDSTNVAFTISMLLGNLNSCCNPWIYMGFNSHLLPRPLRHLACCGGPQ
PRMRRRLSDGSLSSRHTTLLTRSSCPATLSLSLSLTLSGRPRPEESPRDLELADGEGTAE
TIIF
|
|
|
|---|
| BDBM50299343 |
|---|
| n/a |
|---|
| Name | BDBM50299343 |
|---|
| Synonyms: | (2S,4R)-1-((R)-5-chloro-1-(2,4-dimethoxyphenylsulfonyl)-3-(2-methoxyphenyl)-2-oxoindolin-3-yl)-4-hydroxy-N,N-dimethylpyrrolidine-2-carboxamide | (2S,4R)-1-[5-Chloro-1-[(2,4-dimethoxyphenyl)sulfonyl]-3-(2-methoxy-phenyl)-2-oxo-2,3-dihydro-1H-indol-3-yl]-4-hydroxy-N,Ndimethyl-2-pyrrolidine carboxamide | CHEMBL582857 | SR-149415 | SSR149415 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H32ClN3O8S |
|---|
| Mol. Mass. | 630.108 |
|---|
| SMILES | COc1ccc(c(OC)c1)S(=O)(=O)N1C(=O)[C@@](N2C[C@H](O)C[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1ccccc1OC |r| |
|---|
| Structure | 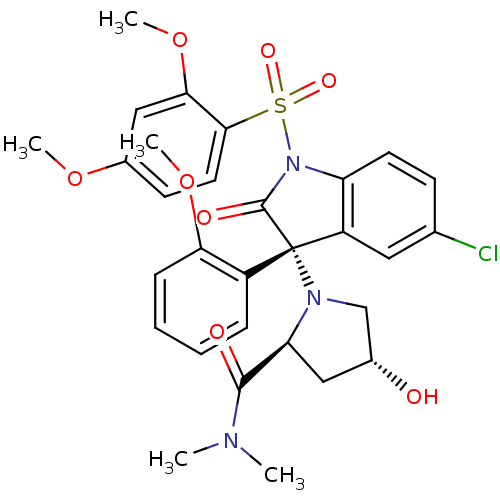
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Scott, JD; Miller, MW; Li, SW; Lin, SI; Vaccaro, HA; Hong, L; Mullins, DE; Guzzi, M; Weinstein, J; Hodgson, RA; Varty, GB; Stamford, AW; Chan, TY; McKittrick, BA; Greenlee, WJ; Priestley, T; Parker, EM Tetrahydroquinoline sulfonamides as vasopressin 1b receptor antagonists. Bioorg Med Chem Lett19:6018-22 (2009) [PubMed] Article
Scott, JD; Miller, MW; Li, SW; Lin, SI; Vaccaro, HA; Hong, L; Mullins, DE; Guzzi, M; Weinstein, J; Hodgson, RA; Varty, GB; Stamford, AW; Chan, TY; McKittrick, BA; Greenlee, WJ; Priestley, T; Parker, EM Tetrahydroquinoline sulfonamides as vasopressin 1b receptor antagonists. Bioorg Med Chem Lett19:6018-22 (2009) [PubMed] Article