| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM50241350 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_633729 (CHEMBL1119100) |
|---|
| IC50 | 5000±n/a nM |
|---|
| Citation |  de Los Ríos, C; Egea, J; Marco-Contelles, J; León, R; Samadi, A; Iriepa, I; Moraleda, I; Gálvez, E; García, AG; López, MG; Villarroya, M; Romero, A Synthesis, inhibitory activity of cholinesterases, and neuroprotective profile of novel 1,8-naphthyridine derivatives. J Med Chem53:5129-43 (2010) [PubMed] Article de Los Ríos, C; Egea, J; Marco-Contelles, J; León, R; Samadi, A; Iriepa, I; Moraleda, I; Gálvez, E; García, AG; López, MG; Villarroya, M; Romero, A Synthesis, inhibitory activity of cholinesterases, and neuroprotective profile of novel 1,8-naphthyridine derivatives. J Med Chem53:5129-43 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | BCHE | Butyrylcholinesterase (BuChE) | CHLE_HORSE | Cholinesterase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65643.35 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | P81908 |
|---|
| Residue: | 574 |
|---|
| Sequence: | EEDIIITTKNGKVRGMNLPVLGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSNIWNATK
YANSCYQNTDQSFPGFLGSEMWNPNTELSEDCLYLNVWIPAPKPKNATVMIWIYGGGFQT
GTSSLPVYDGKFLARVERVIVVSMNYRVGALGFLALSENPEAPGNMGLFDQQLALQWVQK
NIAAFGGNPRSVTLFGESAGAASVSLHLLSPRSQPLFTRAILQSGSSNAPWAVTSLYEAR
NRTLTLAKRMGCSRDNETEMIKCLRDKDPQEILLNEVFVVPYDTLLSVNFGPTVDGDFLT
DMPDTLLQLGQFKRTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPR
VSEFGRESILFHYMDWLDDQRAENYREALDDVVGDYNIICPALEFTRKFSELGNDAFFYY
FEHRSTKLPWPEWMGVMHGYEIEFVFGLPLERRVNYTRAEEILSRSIMKRWANFAKYGNP
NGTQNNSTRWPVFKSTEQKYLTLNTESPKVYTKLRAQQCRFWTLFFPKVLELTGNIDEAE
REWKAGFHRWNNYMMDWKNQFNDYTSKKESCSDF
|
|
|
|---|
| BDBM50241350 |
|---|
| n/a |
|---|
| Name | BDBM50241350 |
|---|
| Synonyms: | 5-Amino-2-methyl-4-phenyl-6,7,8,9-tetrahydro-benzo[b][1,8]naphthyridine-3-carboxylic acid ethyl ester | CHEMBL253386 | ethyl 5-amino-2-methyl-4-phenyl-6,7,8,9-tetrahydrobenzo[b][1,8]naphthyridine-3-carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H23N3O2 |
|---|
| Mol. Mass. | 361.4369 |
|---|
| SMILES | CCOC(=O)c1c(C)nc2nc3CCCCc3c(N)c2c1-c1ccccc1 |
|---|
| Structure | 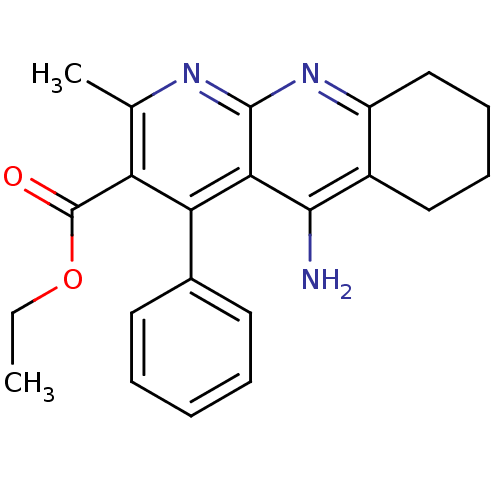
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 de Los Ríos, C; Egea, J; Marco-Contelles, J; León, R; Samadi, A; Iriepa, I; Moraleda, I; Gálvez, E; García, AG; López, MG; Villarroya, M; Romero, A Synthesis, inhibitory activity of cholinesterases, and neuroprotective profile of novel 1,8-naphthyridine derivatives. J Med Chem53:5129-43 (2010) [PubMed] Article
de Los Ríos, C; Egea, J; Marco-Contelles, J; León, R; Samadi, A; Iriepa, I; Moraleda, I; Gálvez, E; García, AG; López, MG; Villarroya, M; Romero, A Synthesis, inhibitory activity of cholinesterases, and neuroprotective profile of novel 1,8-naphthyridine derivatives. J Med Chem53:5129-43 (2010) [PubMed] Article