| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peptidase |
|---|
| Ligand | BDBM50378857 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_705367 (CHEMBL1662792) |
|---|
| pH | 7.4±n/a |
|---|
| IC50 | 1900±n/a nM |
|---|
| Comments | extracted |
|---|
| Citation |  Lemaire, S; Glupczynski, Y; Duval, V; Joris, B; Tulkens, PM; Van Bambeke, F Activities of ceftobiprole and other cephalosporins against extracellular and intracellular (THP-1 macrophages and keratinocytes) forms of methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother53:2289-97 (2009) [PubMed] Article Lemaire, S; Glupczynski, Y; Duval, V; Joris, B; Tulkens, PM; Van Bambeke, F Activities of ceftobiprole and other cephalosporins against extracellular and intracellular (THP-1 macrophages and keratinocytes) forms of methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother53:2289-97 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peptidase |
|---|
| Name: | Peptidase |
|---|
| Synonyms: | MecA | Pbp2a | Penicillin binding protein 2 prime | Penicillin binding protein 2' | Penicillin binding protein 2a (PBP2a) | Penicillin-binding protein 2 prime | Penicillin-binding protein 2' |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 76113.18 |
|---|
| Organism: | Staphylococcus aureus |
|---|
| Description: | ChEMBL_677175 |
|---|
| Residue: | 668 |
|---|
| Sequence: | MKKIKIVPLILIVVVVGFGIYFYASKDKEINNTIDAIEDKNFKQVYKDSSYISKSDNGEV
EMTERPIKIYNSLGVKDINIQDRKIKKVSKNKKRVDAQYKIKTNYGNIDRNVQFNFVKED
GMWKLDWDHSVIIPGMQKDQSIHIENLKSERGKILDRNNVELANTGTAYEIGIVPKNVSK
KDYKAIAKELSISEDYIKQQMDQNWVQDDTFVPLKTVKKMDEYLSDFAKKFHLTTNETES
RNYPLGKATSHLLGYVGPINSEELKQKEYKGYKDDAVIGKKGLEKLYDKKLQHEDGYRVT
IVDDNSNTIAHTLIEKKKKDGKDIQLTIDAKVQKSIYNNMKNDYGSGTAIHPQTGELLAL
VSTPSYDVYPFMYGMSNEEYNKLTEDKKEPLLNKFQITTSPGSTQKILTAMIGLNNKTLD
DKTSYKIDGKGWQKDKSWGGYNVTRYEVVNGNIDLKQAIESSDNIFFARVALELGSKKFE
KGMKKLGVGEDIPSDYPFYNAQISNKNLDNEILLADSGYGQGEILINPVQILSIYSALEN
NGNINAPHLLKDTKNKVWKKNIISKENINLLTDGMQQVVNKTHKEDIYRSYANLIGKSGT
AELKMKQGETGRQIGWFISYDKDNPNMMMAINVKDVQDKGMASYNAKISGKVYDELYENG
NKKYDIDE
|
|
|
|---|
| BDBM50378857 |
|---|
| n/a |
|---|
| Name | BDBM50378857 |
|---|
| Synonyms: | CEFTOBIPROLE | Zeftera | Zevtera |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H22N8O6S2 |
|---|
| Mol. Mass. | 534.569 |
|---|
| SMILES | Nc1nc(ns1)C(N=O)C(=O)N[C@H]1[C@H]2SCC(C=C3CCN([C@@H]4CCNC4)C3=O)=C(N2C1=O)C(O)=O |r,w:17.17,c:31| |
|---|
| Structure | 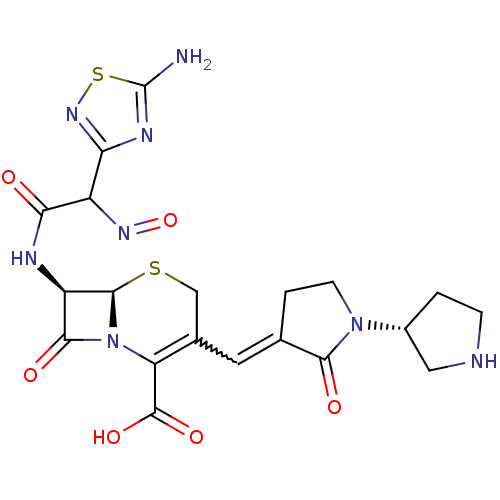
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lemaire, S; Glupczynski, Y; Duval, V; Joris, B; Tulkens, PM; Van Bambeke, F Activities of ceftobiprole and other cephalosporins against extracellular and intracellular (THP-1 macrophages and keratinocytes) forms of methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother53:2289-97 (2009) [PubMed] Article
Lemaire, S; Glupczynski, Y; Duval, V; Joris, B; Tulkens, PM; Van Bambeke, F Activities of ceftobiprole and other cephalosporins against extracellular and intracellular (THP-1 macrophages and keratinocytes) forms of methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother53:2289-97 (2009) [PubMed] Article