| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50380987 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_814102 (CHEMBL2020696) |
|---|
| IC50 | 19000±n/a nM |
|---|
| Citation |  Fournier, PA; Arbour, M; Cauchon, E; Chen, A; Chefson, A; Ducharme, Y; Falgueyret, JP; Gagné, S; Grimm, E; Han, Y; Houle, R; Lacombe, P; Lévesque, JF; MacDonald, D; Mackay, B; McKay, D; Percival, MD; Ramtohul, Y; St-Jacques, R; Toulmond, S Design and synthesis of potent, isoxazole-containing renin inhibitors. Bioorg Med Chem Lett22:2670-4 (2012) [PubMed] Article Fournier, PA; Arbour, M; Cauchon, E; Chen, A; Chefson, A; Ducharme, Y; Falgueyret, JP; Gagné, S; Grimm, E; Han, Y; Houle, R; Lacombe, P; Lévesque, JF; MacDonald, D; Mackay, B; McKay, D; Percival, MD; Ramtohul, Y; St-Jacques, R; Toulmond, S Design and synthesis of potent, isoxazole-containing renin inhibitors. Bioorg Med Chem Lett22:2670-4 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50380987 |
|---|
| n/a |
|---|
| Name | BDBM50380987 |
|---|
| Synonyms: | CHEMBL2017097 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H15BrCl2F2N2O2 |
|---|
| Mol. Mass. | 504.152 |
|---|
| SMILES | O[C@@]1(CCNC[C@@H]1c1onc(c1Br)-c1cccc(Cl)c1Cl)c1ccc(F)c(F)c1 |r| |
|---|
| Structure | 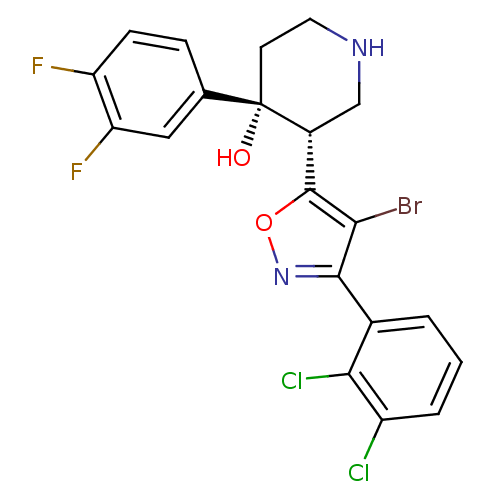
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fournier, PA; Arbour, M; Cauchon, E; Chen, A; Chefson, A; Ducharme, Y; Falgueyret, JP; Gagné, S; Grimm, E; Han, Y; Houle, R; Lacombe, P; Lévesque, JF; MacDonald, D; Mackay, B; McKay, D; Percival, MD; Ramtohul, Y; St-Jacques, R; Toulmond, S Design and synthesis of potent, isoxazole-containing renin inhibitors. Bioorg Med Chem Lett22:2670-4 (2012) [PubMed] Article
Fournier, PA; Arbour, M; Cauchon, E; Chen, A; Chefson, A; Ducharme, Y; Falgueyret, JP; Gagné, S; Grimm, E; Han, Y; Houle, R; Lacombe, P; Lévesque, JF; MacDonald, D; Mackay, B; McKay, D; Percival, MD; Ramtohul, Y; St-Jacques, R; Toulmond, S Design and synthesis of potent, isoxazole-containing renin inhibitors. Bioorg Med Chem Lett22:2670-4 (2012) [PubMed] Article