| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone deacetylase 6 |
|---|
| Ligand | BDBM50386658 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_826886 (CHEMBL2051701) |
|---|
| IC50 | 3.3±n/a nM |
|---|
| Citation |  Lai, MJ; Huang, HL; Pan, SL; Liu, YM; Peng, CY; Lee, HY; Yeh, TK; Huang, PH; Teng, CM; Chen, CS; Chuang, HY; Liou, JP Synthesis and biological evaluation of 1-arylsulfonyl-5-(N-hydroxyacrylamide)indoles as potent histone deacetylase inhibitors with antitumor activity in vivo. J Med Chem55:3777-91 (2012) [PubMed] Article Lai, MJ; Huang, HL; Pan, SL; Liu, YM; Peng, CY; Lee, HY; Yeh, TK; Huang, PH; Teng, CM; Chen, CS; Chuang, HY; Liou, JP Synthesis and biological evaluation of 1-arylsulfonyl-5-(N-hydroxyacrylamide)indoles as potent histone deacetylase inhibitors with antitumor activity in vivo. J Med Chem55:3777-91 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone deacetylase 6 |
|---|
| Name: | Histone deacetylase 6 |
|---|
| Synonyms: | Cereblon/Histone deacetylase 6 | HD6 | HDAC6 | HDAC6_HUMAN | Histone deacetylase 6 (HDAC6) | Human HDAC6 | KIAA0901 | ORF Names:JM21 |
|---|
| Type: | Chromatin regulator; hydrolase; repressor |
|---|
| Mol. Mass.: | 131381.51 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9UBN7 |
|---|
| Residue: | 1215 |
|---|
| Sequence: | MTSTGQDSTTTRQRRSRQNPQSPPQDSSVTSKRNIKKGAVPRSIPNLAEVKKKGKMKKLG
QAMEEDLIVGLQGMDLNLEAEALAGTGLVLDEQLNEFHCLWDDSFPEGPERLHAIKEQLI
QEGLLDRCVSFQARFAEKEELMLVHSLEYIDLMETTQYMNEGELRVLADTYDSVYLHPNS
YSCACLASGSVLRLVDAVLGAEIRNGMAIIRPPGHHAQHSLMDGYCMFNHVAVAARYAQQ
KHRIRRVLIVDWDVHHGQGTQFTFDQDPSVLYFSIHRYEQGRFWPHLKASNWSTTGFGQG
QGYTINVPWNQVGMRDADYIAAFLHVLLPVALEFQPQLVLVAAGFDALQGDPKGEMAATP
AGFAQLTHLLMGLAGGKLILSLEGGYNLRALAEGVSASLHTLLGDPCPMLESPGAPCRSA
QASVSCALEALEPFWEVLVRSTETVERDNMEEDNVEESEEEGPWEPPVLPILTWPVLQSR
TGLVYDQNMMNHCNLWDSHHPEVPQRILRIMCRLEELGLAGRCLTLTPRPATEAELLTCH
SAEYVGHLRATEKMKTRELHRESSNFDSIYICPSTFACAQLATGAACRLVEAVLSGEVLN
GAAVVRPPGHHAEQDAACGFCFFNSVAVAARHAQTISGHALRILIVDWDVHHGNGTQHMF
EDDPSVLYVSLHRYDHGTFFPMGDEGASSQIGRAAGTGFTVNVAWNGPRMGDADYLAAWH
RLVLPIAYEFNPELVLVSAGFDAARGDPLGGCQVSPEGYAHLTHLLMGLASGRIILILEG
GYNLTSISESMAACTRSLLGDPPPLLTLPRPPLSGALASITETIQVHRRYWRSLRVMKVE
DREGPSSSKLVTKKAPQPAKPRLAERMTTREKKVLEAGMGKVTSASFGEESTPGQTNSET
AVVALTQDQPSEAATGGATLAQTISEAAIGGAMLGQTTSEEAVGGATPDQTTSEETVGGA
ILDQTTSEDAVGGATLGQTTSEEAVGGATLAQTTSEAAMEGATLDQTTSEEAPGGTELIQ
TPLASSTDHQTPPTSPVQGTTPQISPSTLIGSLRTLELGSESQGASESQAPGEENLLGEA
AGGQDMADSMLMQGSRGLTDQAIFYAVTPLPWCPHLVAVCPIPAAGLDVTQPCGDCGTIQ
ENWVCLSCYQVYCGRYINGHMLQHHGNSGHPLVLSYIDLSAWCYYCQAYVHHQALLDVKN
IAHQNKFGEDMPHPH
|
|
|
|---|
| BDBM50386658 |
|---|
| n/a |
|---|
| Name | BDBM50386658 |
|---|
| Synonyms: | CHEMBL2048749 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H16N2O5S |
|---|
| Mol. Mass. | 372.395 |
|---|
| SMILES | COc1ccc(cc1)S(=O)(=O)n1ccc2cc(\C=C\C(=O)NO)ccc12 |
|---|
| Structure | 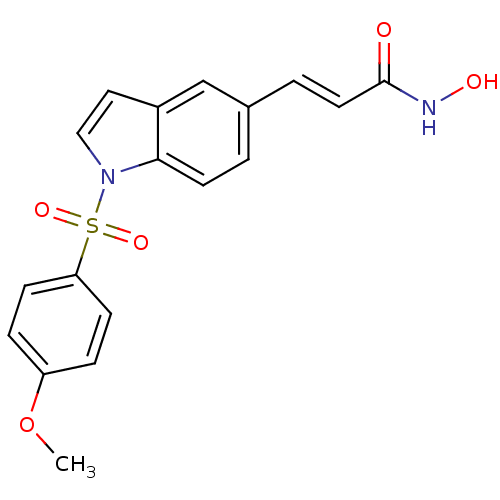
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lai, MJ; Huang, HL; Pan, SL; Liu, YM; Peng, CY; Lee, HY; Yeh, TK; Huang, PH; Teng, CM; Chen, CS; Chuang, HY; Liou, JP Synthesis and biological evaluation of 1-arylsulfonyl-5-(N-hydroxyacrylamide)indoles as potent histone deacetylase inhibitors with antitumor activity in vivo. J Med Chem55:3777-91 (2012) [PubMed] Article
Lai, MJ; Huang, HL; Pan, SL; Liu, YM; Peng, CY; Lee, HY; Yeh, TK; Huang, PH; Teng, CM; Chen, CS; Chuang, HY; Liou, JP Synthesis and biological evaluation of 1-arylsulfonyl-5-(N-hydroxyacrylamide)indoles as potent histone deacetylase inhibitors with antitumor activity in vivo. J Med Chem55:3777-91 (2012) [PubMed] Article