| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sodium/glucose cotransporter 1 |
|---|
| Ligand | BDBM50335145 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_831838 (CHEMBL2065402) |
|---|
| EC50 | 364±n/a nM |
|---|
| Citation |  Yao, CH; Song, JS; Chen, CT; Yeh, TK; Hsieh, TC; Wu, SH; Huang, CY; Huang, YL; Wang, MH; Liu, YW; Tsai, CH; Kumar, CR; Lee, JC Synthesis and biological evaluation of novel C-indolylxylosides as sodium-dependent glucose co-transporter 2 inhibitors. Eur J Med Chem55:32-8 (2012) [PubMed] Article Yao, CH; Song, JS; Chen, CT; Yeh, TK; Hsieh, TC; Wu, SH; Huang, CY; Huang, YL; Wang, MH; Liu, YW; Tsai, CH; Kumar, CR; Lee, JC Synthesis and biological evaluation of novel C-indolylxylosides as sodium-dependent glucose co-transporter 2 inhibitors. Eur J Med Chem55:32-8 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sodium/glucose cotransporter 1 |
|---|
| Name: | Sodium/glucose cotransporter 1 |
|---|
| Synonyms: | High affinity sodium-glucose cotransporter | NAGT | Na(+)/glucose cotransporter 1 | SC5A1_HUMAN | SGLT1 | SLC5A1 | Sodium-dependent glucose cotransporter 1 (SGLT1) | Sodium/glucose cotransporter 1 | Sodium/glucose cotransporter 1 (SGLT1) | Solute carrier family 5 member 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 73503.78 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P13866 |
|---|
| Residue: | 664 |
|---|
| Sequence: | MDSSTWSPKTTAVTRPVETHELIRNAADISIIVIYFVVVMAVGLWAMFSTNRGTVGGFFL
AGRSMVWWPIGASLFASNIGSGHFVGLAGTGAASGIAIGGFEWNALVLVVVLGWLFVPIY
IKAGVVTMPEYLRKRFGGQRIQVYLSLLSLLLYIFTKISADIFSGAIFINLALGLNLYLA
IFLLLAITALYTITGGLAAVIYTDTLQTVIMLVGSLILTGFAFHEVGGYDAFMEKYMKAI
PTIVSDGNTTFQEKCYTPRADSFHIFRDPLTGDLPWPGFIFGMSILTLWYWCTDQVIVQR
CLSAKNMSHVKGGCILCGYLKLMPMFIMVMPGMISRILYTEKIACVVPSECEKYCGTKVG
CTNIAYPTLVVELMPNGLRGLMLSVMLASLMSSLTSIFNSASTLFTMDIYAKVRKRASEK
ELMIAGRLFILVLIGISIAWVPIVQSAQSGQLFDYIQSITSYLGPPIAAVFLLAIFWKRV
NEPGAFWGLILGLLIGISRMITEFAYGTGSCMEPSNCPTIICGVHYLYFAIILFAISFIT
IVVISLLTKPIPDVHLYRLCWSLRNSKEERIDLDAEEENIQEGPKETIEIETQVPEKKKG
IFRRAYDLFCGLEQHGAPKMTEEEEKAMKMKMTDTSEKPLWRTVLNVNGIILVTVAVFCH
AYFA
|
|
|
|---|
| BDBM50335145 |
|---|
| n/a |
|---|
| Name | BDBM50335145 |
|---|
| Synonyms: | 4-Chloro-3-(4-cyclopropylbenzyl)-1-(beta-D-xylopyranosyl)-1Hindole | CHEMBL1650501 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H24ClNO4 |
|---|
| Mol. Mass. | 413.894 |
|---|
| SMILES | O[C@@H]1CO[C@H]([C@H](O)[C@H]1O)n1cc(Cc2ccc(cc2)C2CC2)c2c(Cl)cccc12 |r| |
|---|
| Structure | 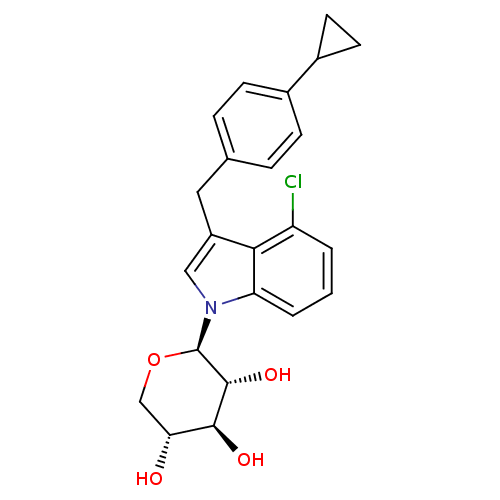
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yao, CH; Song, JS; Chen, CT; Yeh, TK; Hsieh, TC; Wu, SH; Huang, CY; Huang, YL; Wang, MH; Liu, YW; Tsai, CH; Kumar, CR; Lee, JC Synthesis and biological evaluation of novel C-indolylxylosides as sodium-dependent glucose co-transporter 2 inhibitors. Eur J Med Chem55:32-8 (2012) [PubMed] Article
Yao, CH; Song, JS; Chen, CT; Yeh, TK; Hsieh, TC; Wu, SH; Huang, CY; Huang, YL; Wang, MH; Liu, YW; Tsai, CH; Kumar, CR; Lee, JC Synthesis and biological evaluation of novel C-indolylxylosides as sodium-dependent glucose co-transporter 2 inhibitors. Eur J Med Chem55:32-8 (2012) [PubMed] Article