| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Diacylglycerol O-acyltransferase 1 |
|---|
| Ligand | BDBM50399715 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_876789 (CHEMBL2183606) |
|---|
| IC50 | 40±n/a nM |
|---|
| Citation |  Barlind, JG; Bauer, UA; Birch, AM; Birtles, S; Buckett, LK; Butlin, RJ; Davies, RD; Eriksson, JW; Hammond, CD; Hovland, R; Johannesson, P; Johansson, MJ; Kemmitt, PD; Lindmark, BT; Morentin Gutierrez, P; Noeske, TA; Nordin, A; O'Donnell, CJ; Petersson, AU; Redzic, A; Turnbull, AV; Vinblad, J Design and optimization of pyrazinecarboxamide-based inhibitors of diacylglycerol acyltransferase 1 (DGAT1) leading to a clinical candidate dimethylpyrazinecarboxamide phenylcyclohexylacetic acid (AZD7687). J Med Chem55:10610-29 (2012) [PubMed] Article Barlind, JG; Bauer, UA; Birch, AM; Birtles, S; Buckett, LK; Butlin, RJ; Davies, RD; Eriksson, JW; Hammond, CD; Hovland, R; Johannesson, P; Johansson, MJ; Kemmitt, PD; Lindmark, BT; Morentin Gutierrez, P; Noeske, TA; Nordin, A; O'Donnell, CJ; Petersson, AU; Redzic, A; Turnbull, AV; Vinblad, J Design and optimization of pyrazinecarboxamide-based inhibitors of diacylglycerol acyltransferase 1 (DGAT1) leading to a clinical candidate dimethylpyrazinecarboxamide phenylcyclohexylacetic acid (AZD7687). J Med Chem55:10610-29 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Diacylglycerol O-acyltransferase 1 |
|---|
| Name: | Diacylglycerol O-acyltransferase 1 |
|---|
| Synonyms: | ACAT-related gene product 1 | AGRP1 | Acyl coA-diacylglycerol acyl transferase 1 (DGAT1) | DGAT | DGAT1 | DGAT1_HUMAN | Diacylglycerol Acyltransferase (DGAT1) | Diacylglycerol O-acyltransferase 1 | Diacylglycerol O-acyltransferase 1 (DGAT1) | Diglyceride acyltransferase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55297.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O75907 |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGDRGSSRRRRTGSRPSSHGGGGPAAAEEEVRDAAAGPDVGAAGDAPAPAPNKDGDAGVG
SGHWELRCHRLQDSLFSSDSGFSNYRGILNWCVVMLILSNARLFLENLIKYGILVDPIQV
VSLFLKDPYSWPAPCLVIAANVFAVAAFQVEKRLAVGALTEQAGLLLHVANLATILCFPA
AVVLLVESITPVGSLLALMAHTILFLKLFSYRDVNSWCRRARAKAASAGKKASSAAAPHT
VSYPDNLTYRDLYYFLFAPTLCYELNFPRSPRIRKRFLLRRILEMLFFTQLQVGLIQQWM
VPTIQNSMKPFKDMDYSRIIERLLKLAVPNHLIWLIFFYWLFHSCLNAVAELMQFGDREF
YRDWWNSESVTYFWQNWNIPVHKWCIRHFYKPMLRRGSSKWMARTGVFLASAFFHEYLVS
VPLRMFRLWAFTGMMAQIPLAWFVGRFFQGNYGNAAVWLSLIIGQPIAVLMYVHDYYVLN
YEAPAAEA
|
|
|
|---|
| BDBM50399715 |
|---|
| n/a |
|---|
| Name | BDBM50399715 |
|---|
| Synonyms: | CHEMBL2178373 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H27N7O |
|---|
| Mol. Mass. | 417.5068 |
|---|
| SMILES | Cc1nc(C)c(nc1C(N)=O)-c1ccc(cc1)C12CCC(Cc3nnn[nH]3)(CC1)CC2 |
|---|
| Structure | 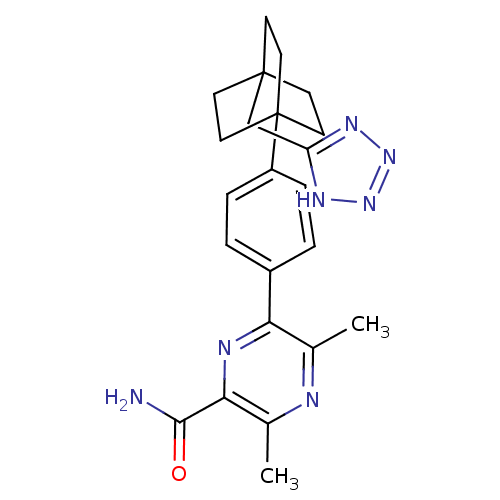
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Barlind, JG; Bauer, UA; Birch, AM; Birtles, S; Buckett, LK; Butlin, RJ; Davies, RD; Eriksson, JW; Hammond, CD; Hovland, R; Johannesson, P; Johansson, MJ; Kemmitt, PD; Lindmark, BT; Morentin Gutierrez, P; Noeske, TA; Nordin, A; O'Donnell, CJ; Petersson, AU; Redzic, A; Turnbull, AV; Vinblad, J Design and optimization of pyrazinecarboxamide-based inhibitors of diacylglycerol acyltransferase 1 (DGAT1) leading to a clinical candidate dimethylpyrazinecarboxamide phenylcyclohexylacetic acid (AZD7687). J Med Chem55:10610-29 (2012) [PubMed] Article
Barlind, JG; Bauer, UA; Birch, AM; Birtles, S; Buckett, LK; Butlin, RJ; Davies, RD; Eriksson, JW; Hammond, CD; Hovland, R; Johannesson, P; Johansson, MJ; Kemmitt, PD; Lindmark, BT; Morentin Gutierrez, P; Noeske, TA; Nordin, A; O'Donnell, CJ; Petersson, AU; Redzic, A; Turnbull, AV; Vinblad, J Design and optimization of pyrazinecarboxamide-based inhibitors of diacylglycerol acyltransferase 1 (DGAT1) leading to a clinical candidate dimethylpyrazinecarboxamide phenylcyclohexylacetic acid (AZD7687). J Med Chem55:10610-29 (2012) [PubMed] Article