| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M2 |
|---|
| Ligand | BDBM50074569 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_139918 (CHEMBL748586) |
|---|
| EC50 | 1412.54±n/a nM |
|---|
| Citation |  Botero Cid, HM; Tränkle, C; Baumann, K; Pick, R; Mies-Klomfass, E; Kostenis, E; Mohr, K; Holzgrabe, U Structure-activity relationships in a series of bisquaternary bisphthalimidine derivatives modulating the muscarinic M(2)-receptor allosterically. J Med Chem43:2155-64 (2000) [PubMed] Botero Cid, HM; Tränkle, C; Baumann, K; Pick, R; Mies-Klomfass, E; Kostenis, E; Mohr, K; Holzgrabe, U Structure-activity relationships in a series of bisquaternary bisphthalimidine derivatives modulating the muscarinic M(2)-receptor allosterically. J Med Chem43:2155-64 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Muscarinic acetylcholine receptor M2 |
|---|
| Name: | Muscarinic acetylcholine receptor M2 |
|---|
| Synonyms: | ACM2_PIG | CHRM2 | Cholinergic, muscarinic | GPM2 | LOC100715343 | Muscarinic acetylcholine receptor M2 | Muscarinic receptor 2 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 51688.65 |
|---|
| Organism: | GUINEA PIG |
|---|
| Description: | Cholinergic, muscarinic 0 GUINEA PIG::P06199 |
|---|
| Residue: | 466 |
|---|
| Sequence: | MNNSTNSSNSGLALTSPYKTFEVVFIVLVAGSLSLVTIIGNILVMVSIKVNRHLQTVNNY
FLFSLACADLIIGVFSMNLYTLYTVIGYWPLGPVVCDLWLALDYVVSNASVMNLLIISFD
RYFCVTKPLTYPVKRTTKMAGMMIAAAWVLSFILWAPAILFWQFIVGVRTVEDGECYIQF
FSNAAVTFGTAIAAFYLPVIIMTVLYWHISRASKSRIKKDKKEPVANQEPVSPSLVQGRI
VKPNNNNMPGSDEALEHNKIQNGKAPRDAVTENCVQGEEKESSNDSTSVSAVASNMRDDE
ITQDENTVSTSLGHSKDENSKQTCIKIVTKTQKSDSCTPANTTVELVGSSGQNGDEKQNI
VARKIVKMTKQPAKKKPPPSREKKVTRTILAILLAFIITWAPYNVMVLINTFCAPCIPNT
VWTIGYWLCYINSTINPACYALCNATFKKTFKHLLMCHYKNIGATR
|
|
|
|---|
| BDBM50074569 |
|---|
| n/a |
|---|
| Name | BDBM50074569 |
|---|
| Synonyms: | (6-{bis[3-(1,3-Dioxo-1,3-dihydro-isoindol-2-yl)-propyl]-methyl-ammonio}-hexyl)-dimethyl-ammonium Dibromide | Bis (ammonio) alkane analogue | Bis-{3-(1,3-dioxo-2,3-dihydro-1H-2-isoindolyl)propyl(trimethyl)ammonium}butane; dibromide | CHEMBL266295 | N1,N6-bis(3-(1,3-dioxoisoindolin-2-yl)propyl)-N1,N1,N6,N6-tetramethylhexane-1,6-diaminium bromide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H44N4O4 |
|---|
| Mol. Mass. | 548.7151 |
|---|
| SMILES | C[N+](C)(CCCCCC[N+](C)(C)CCCN1C(=O)c2ccccc2C1=O)CCCN1C(=O)c2ccccc2C1=O |
|---|
| Structure | 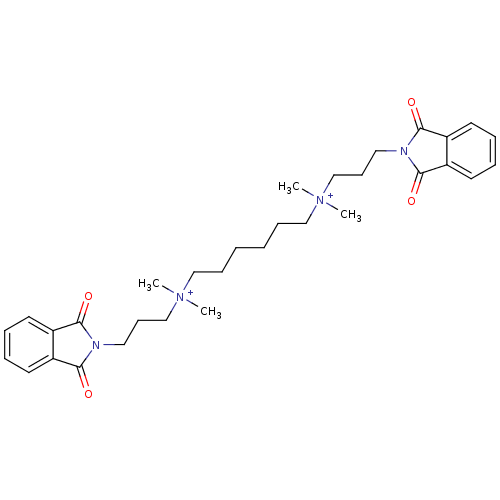
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Botero Cid, HM; Tränkle, C; Baumann, K; Pick, R; Mies-Klomfass, E; Kostenis, E; Mohr, K; Holzgrabe, U Structure-activity relationships in a series of bisquaternary bisphthalimidine derivatives modulating the muscarinic M(2)-receptor allosterically. J Med Chem43:2155-64 (2000) [PubMed]
Botero Cid, HM; Tränkle, C; Baumann, K; Pick, R; Mies-Klomfass, E; Kostenis, E; Mohr, K; Holzgrabe, U Structure-activity relationships in a series of bisquaternary bisphthalimidine derivatives modulating the muscarinic M(2)-receptor allosterically. J Med Chem43:2155-64 (2000) [PubMed]