| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM21392 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_201057 (CHEMBL805833) |
|---|
| IC50 | 21878±n/a nM |
|---|
| Citation |  Russell, MG; Castro, JL; Matassa, VG; Beer, MS; Heald, A; Scholey, K; Stanton, JA; Broughton, HB Synthesis and serotonergic activity of benzofuran and dihydrogenzofuran analogues of 5-carboxamidotryptamine. Bioorg Med Chem Lett4:1207-1212 (1994) Article Russell, MG; Castro, JL; Matassa, VG; Beer, MS; Heald, A; Scholey, K; Stanton, JA; Broughton, HB Synthesis and serotonergic activity of benzofuran and dihydrogenzofuran analogues of 5-carboxamidotryptamine. Bioorg Med Chem Lett4:1207-1212 (1994) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52852.05 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cortex membranes 5-HT2A receptors. |
|---|
| Residue: | 471 |
|---|
| Sequence: | MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGY
LPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYK
SSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
|
|
|
|---|
| BDBM21392 |
|---|
| n/a |
|---|
| Name | BDBM21392 |
|---|
| Synonyms: | 3-(2-aminoethyl)-1H-indole-5-carboxamide | 5-CT | 5-CT,DP | 5-carboxamidotryptamine | CHEMBL1256863 | CHEMBL18840 | [3H]-5-CT | [3H]-5-Carboxamide tryptamine | [3H]-5-Carboxyamidotryptamine | [3H]-5-carboxamidotryptamine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H13N3O |
|---|
| Mol. Mass. | 203.2404 |
|---|
| SMILES | NCCc1c[nH]c2ccc(cc12)C(N)=O |
|---|
| Structure | 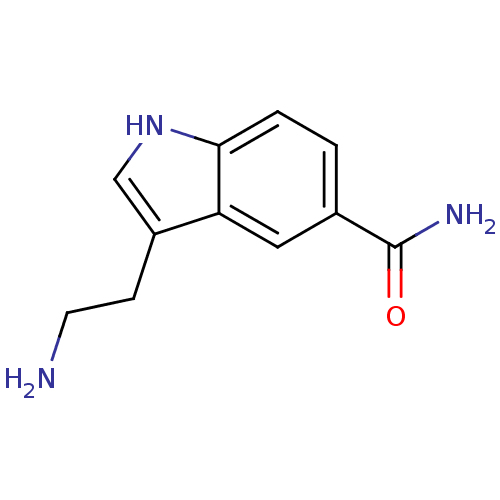
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Russell, MG; Castro, JL; Matassa, VG; Beer, MS; Heald, A; Scholey, K; Stanton, JA; Broughton, HB Synthesis and serotonergic activity of benzofuran and dihydrogenzofuran analogues of 5-carboxamidotryptamine. Bioorg Med Chem Lett4:1207-1212 (1994) Article
Russell, MG; Castro, JL; Matassa, VG; Beer, MS; Heald, A; Scholey, K; Stanton, JA; Broughton, HB Synthesis and serotonergic activity of benzofuran and dihydrogenzofuran analogues of 5-carboxamidotryptamine. Bioorg Med Chem Lett4:1207-1212 (1994) Article