| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50429052 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_942279 (CHEMBL2343277) |
|---|
| Ki | 363±n/a nM |
|---|
| Citation |  Sun, H; Zhu, L; Yang, H; Qian, W; Guo, L; Zhou, S; Gao, B; Li, Z; Zhou, Y; Jiang, H; Chen, K; Zhen, X; Liu, H Asymmetric total synthesis and identification of tetrahydroprotoberberine derivatives as new antipsychotic agents possessing a dopamine D(1), D(2) and serotonin 5-HT(1A) multi-action profile. Bioorg Med Chem21:856-68 (2013) [PubMed] Article Sun, H; Zhu, L; Yang, H; Qian, W; Guo, L; Zhou, S; Gao, B; Li, Z; Zhou, Y; Jiang, H; Chen, K; Zhen, X; Liu, H Asymmetric total synthesis and identification of tetrahydroprotoberberine derivatives as new antipsychotic agents possessing a dopamine D(1), D(2) and serotonin 5-HT(1A) multi-action profile. Bioorg Med Chem21:856-68 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM50429052 |
|---|
| n/a |
|---|
| Name | BDBM50429052 |
|---|
| Synonyms: | CHEMBL2334881 | US9359372, DC037082 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H21NO4 |
|---|
| Mol. Mass. | 339.385 |
|---|
| SMILES | COc1c(O)ccc2C[C@@H]3N(CCc4cc5OCCOc5cc34)Cc12 |r| |
|---|
| Structure | 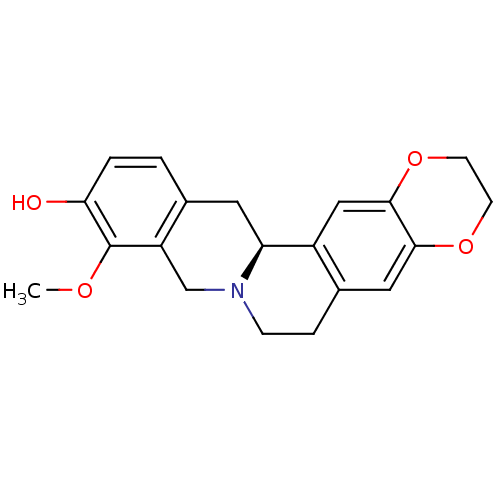
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sun, H; Zhu, L; Yang, H; Qian, W; Guo, L; Zhou, S; Gao, B; Li, Z; Zhou, Y; Jiang, H; Chen, K; Zhen, X; Liu, H Asymmetric total synthesis and identification of tetrahydroprotoberberine derivatives as new antipsychotic agents possessing a dopamine D(1), D(2) and serotonin 5-HT(1A) multi-action profile. Bioorg Med Chem21:856-68 (2013) [PubMed] Article
Sun, H; Zhu, L; Yang, H; Qian, W; Guo, L; Zhou, S; Gao, B; Li, Z; Zhou, Y; Jiang, H; Chen, K; Zhen, X; Liu, H Asymmetric total synthesis and identification of tetrahydroprotoberberine derivatives as new antipsychotic agents possessing a dopamine D(1), D(2) and serotonin 5-HT(1A) multi-action profile. Bioorg Med Chem21:856-68 (2013) [PubMed] Article