| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50441232 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_984534 (CHEMBL2433740) |
|---|
| IC50 | 5300±n/a nM |
|---|
| Citation |  Turlington, M; Noetzel, MJ; Chun, A; Zhou, Y; Gogliotti, RD; Nguyen, ED; Gregory, KJ; Vinson, PN; Rook, JM; Gogi, KK; Xiang, Z; Bridges, TM; Daniels, JS; Jones, C; Niswender, CM; Meiler, J; Conn, PJ; Lindsley, CW; Stauffer, SR Exploration of allosteric agonism structure-activity relationships within an acetylene series of metabotropic glutamate receptor 5 (mGlu5) positive allosteric modulators (PAMs): discovery of 5-((3-fluorophenyl)ethynyl)-N-(3-methyloxetan-3-yl)picolinamide (ML254). J Med Chem56:7976-96 (2013) [PubMed] Article Turlington, M; Noetzel, MJ; Chun, A; Zhou, Y; Gogliotti, RD; Nguyen, ED; Gregory, KJ; Vinson, PN; Rook, JM; Gogi, KK; Xiang, Z; Bridges, TM; Daniels, JS; Jones, C; Niswender, CM; Meiler, J; Conn, PJ; Lindsley, CW; Stauffer, SR Exploration of allosteric agonism structure-activity relationships within an acetylene series of metabotropic glutamate receptor 5 (mGlu5) positive allosteric modulators (PAMs): discovery of 5-((3-fluorophenyl)ethynyl)-N-(3-methyloxetan-3-yl)picolinamide (ML254). J Med Chem56:7976-96 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50441232 |
|---|
| n/a |
|---|
| Name | BDBM50441232 |
|---|
| Synonyms: | CHEMBL2431173 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H15FN2O2 |
|---|
| Mol. Mass. | 310.3223 |
|---|
| SMILES | CC1(COC1)NC(=O)c1ccc(cn1)C#Cc1cccc(F)c1 |
|---|
| Structure | 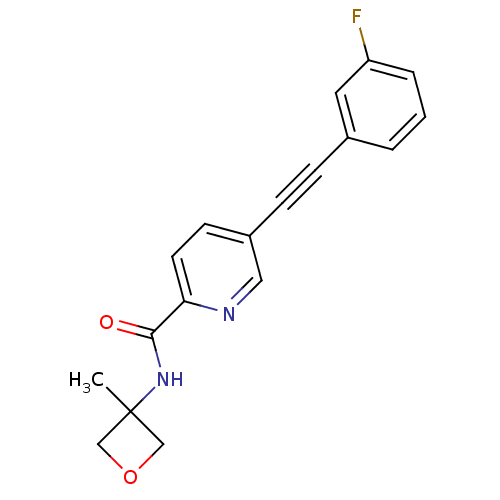
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Turlington, M; Noetzel, MJ; Chun, A; Zhou, Y; Gogliotti, RD; Nguyen, ED; Gregory, KJ; Vinson, PN; Rook, JM; Gogi, KK; Xiang, Z; Bridges, TM; Daniels, JS; Jones, C; Niswender, CM; Meiler, J; Conn, PJ; Lindsley, CW; Stauffer, SR Exploration of allosteric agonism structure-activity relationships within an acetylene series of metabotropic glutamate receptor 5 (mGlu5) positive allosteric modulators (PAMs): discovery of 5-((3-fluorophenyl)ethynyl)-N-(3-methyloxetan-3-yl)picolinamide (ML254). J Med Chem56:7976-96 (2013) [PubMed] Article
Turlington, M; Noetzel, MJ; Chun, A; Zhou, Y; Gogliotti, RD; Nguyen, ED; Gregory, KJ; Vinson, PN; Rook, JM; Gogi, KK; Xiang, Z; Bridges, TM; Daniels, JS; Jones, C; Niswender, CM; Meiler, J; Conn, PJ; Lindsley, CW; Stauffer, SR Exploration of allosteric agonism structure-activity relationships within an acetylene series of metabotropic glutamate receptor 5 (mGlu5) positive allosteric modulators (PAMs): discovery of 5-((3-fluorophenyl)ethynyl)-N-(3-methyloxetan-3-yl)picolinamide (ML254). J Med Chem56:7976-96 (2013) [PubMed] Article