| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuropeptide Y receptor type 5 |
|---|
| Ligand | BDBM50441965 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_992052 (CHEMBL2447453) |
|---|
| Ki | 17±n/a nM |
|---|
| Citation |  Berlicki, L; Kaske, M; Gutièrrez-Abad, R; Bernhardt, G; Illa, O; Ortuno, RM; Cabrele, C; Buschauer, A; Reiser, O Replacement of Thr32 and Gln34 in the C-terminal neuropeptide Y fragment 25-36 by cis-cyclobutane and cis-cyclopentane β-amino acids shifts selectivity toward the Y(4) receptor. J Med Chem56:8422-31 (2013) [PubMed] Article Berlicki, L; Kaske, M; Gutièrrez-Abad, R; Bernhardt, G; Illa, O; Ortuno, RM; Cabrele, C; Buschauer, A; Reiser, O Replacement of Thr32 and Gln34 in the C-terminal neuropeptide Y fragment 25-36 by cis-cyclobutane and cis-cyclopentane β-amino acids shifts selectivity toward the Y(4) receptor. J Med Chem56:8422-31 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuropeptide Y receptor type 5 |
|---|
| Name: | Neuropeptide Y receptor type 5 |
|---|
| Synonyms: | NPY-Y5 | NPY-Y5 receptor | NPY5-R | NPY5R | NPY5R_HUMAN | NPYR5 | NPYY5 | Neuropeptide Y receptor type 5 | Neuropeptide Y receptor type 5 ( NPY Y5) | Y5 receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 50746.64 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15761 |
|---|
| Residue: | 445 |
|---|
| Sequence: | MDLELDEYYNKTLATENNTAATRNSDFPVWDDYKSSVDDLQYFLIGLYTFVSLLGFMGNL
LILMALMKKRNQKTTVNFLIGNLAFSDILVVLFCSPFTLTSVLLDQWMFGKVMCHIMPFL
QCVSVLVSTLILISIAIVRYHMIKHPISNNLTANHGYFLIATVWTLGFAICSPLPVFHSL
VELQETFGSALLSSRYLCVESWPSDSYRIAFTISLLLVQYILPLVCLTVSHTSVCRSISC
GLSNKENRLEENEMINLTLHPSKKSGPQVKLSGSHKWSYSFIKKHRRRYSKKTACVLPAP
ERPSQENHSRILPENFGSVRSQLSSSSKFIPGVPTCFEIKPEENSDVHELRVKRSVTRIK
KRSRSVFYRLTILILVFAVSWMPLHLFHVVTDFNDNLISNRHFKLVYCICHLLGMMSCCL
NPILYGFLNNGIKADLVSLIHCLHM
|
|
|
|---|
| BDBM50441965 |
|---|
| n/a |
|---|
| Name | BDBM50441965 |
|---|
| Synonyms: | CHEMBL2440192 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C184H287N53O53S2 |
|---|
| Mol. Mass. | 4153.701 |
|---|
| SMILES | [#6]-[#6]-[#6@H](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#16]-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#16]-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6])-[#7])-[#6](-[#6])-[#6])-[#6@@H](-[#6])-[#8])-[#6@@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7])-[#6](=O)-[#7]-[#6@H](-[#7])-[#6]-c1ccc(-[#8])cc1 |r| |
|---|
| Structure | 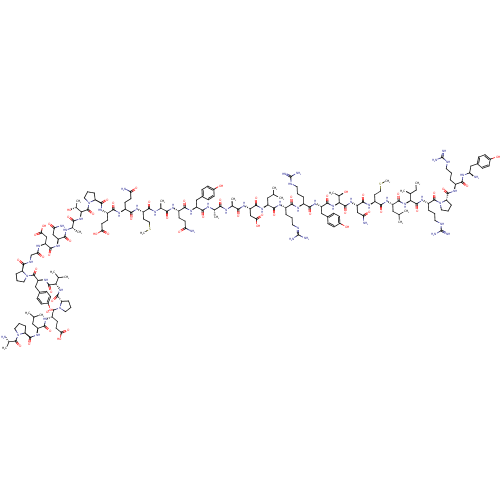
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Berlicki, L; Kaske, M; Gutièrrez-Abad, R; Bernhardt, G; Illa, O; Ortuno, RM; Cabrele, C; Buschauer, A; Reiser, O Replacement of Thr32 and Gln34 in the C-terminal neuropeptide Y fragment 25-36 by cis-cyclobutane and cis-cyclopentane β-amino acids shifts selectivity toward the Y(4) receptor. J Med Chem56:8422-31 (2013) [PubMed] Article
Berlicki, L; Kaske, M; Gutièrrez-Abad, R; Bernhardt, G; Illa, O; Ortuno, RM; Cabrele, C; Buschauer, A; Reiser, O Replacement of Thr32 and Gln34 in the C-terminal neuropeptide Y fragment 25-36 by cis-cyclobutane and cis-cyclopentane β-amino acids shifts selectivity toward the Y(4) receptor. J Med Chem56:8422-31 (2013) [PubMed] Article