| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glucocorticoid receptor |
|---|
| Ligand | BDBM50048341 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1459138 (CHEMBL3370899) |
|---|
| EC50 | 11±n/a nM |
|---|
| Citation |  Gong, H; Yang, M; Xiao, Z; Doweyko, AM; Cunningham, M; Wang, J; Habte, S; Holloway, D; Burke, C; Shuster, D; Gao, L; Carman, J; Somerville, JE; Nadler, SG; Salter-Cid, L; Barrish, JC; Weinstein, DS Discovery of acylurea isosteres of 2-acylaminothiadiazole in the azaxanthene series of glucocorticoid receptor agonists. Bioorg Med Chem Lett24:3268-73 (2014) [PubMed] Article Gong, H; Yang, M; Xiao, Z; Doweyko, AM; Cunningham, M; Wang, J; Habte, S; Holloway, D; Burke, C; Shuster, D; Gao, L; Carman, J; Somerville, JE; Nadler, SG; Salter-Cid, L; Barrish, JC; Weinstein, DS Discovery of acylurea isosteres of 2-acylaminothiadiazole in the azaxanthene series of glucocorticoid receptor agonists. Bioorg Med Chem Lett24:3268-73 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glucocorticoid receptor |
|---|
| Name: | Glucocorticoid receptor |
|---|
| Synonyms: | GCR_HUMAN | GR | GRL | Glucocorticoid | Glucocorticoid receptor (GRFP) | NR3C1 | Nuclear receptor subfamily 3 group C member 1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 85656.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04150 |
|---|
| Residue: | 777 |
|---|
| Sequence: | MDSKESLTPGREENPSSVLAQERGDVMDFYKTLRGGATVKVSASSPSLAVASQSDSKQRR
LLVDFPKGSVSNAQQPDLSKAVSLSMGLYMGETETKVMGNDLGFPQQGQISLSSGETDLK
LLEESIANLNRSTSVPENPKSSASTAVSAAPTEKEFPKTHSDVSSEQQHLKGQTGTNGGN
VKLYTTDQSTFDILQDLEFSSGSPGKETNESPWRSDLLIDENCLLSPLAGEDDSFLLEGN
SNEDCKPLILPDTKPKIKDNGDLVLSSPSNVTLPQVKTEKEDFIELCTPGVIKQEKLGTV
YCQASFPGANIIGNKMSAISVHGVSTSGGQMYHYDMNTASLSQQQDQKPIFNVIPPIPVG
SENWNRCQGSGDDNLTSLGTLNFPGRTVFSNGYSSPSMRPDVSSPPSSSSTATTGPPPKL
CLVCSDEASGCHYGVLTCGSCKVFFKRAVEGQHNYLCAGRNDCIIDKIRRKNCPACRYRK
CLQAGMNLEARKTKKKIKGIQQATTGVSQETSENPGNKTIVPATLPQLTPTLVSLLEVIE
PEVLYAGYDSSVPDSTWRIMTTLNMLGGRQVIAAVKWAKAIPGFRNLHLDDQMTLLQYSW
MFLMAFALGWRSYRQSSANLLCFAPDLIINEQRMTLPCMYDQCKHMLYVSSELHRLQVSY
EEYLCMKTLLLLSSVPKDGLKSQELFDEIRMTYIKELGKAIVKREGNSSQNWQRFYQLTK
LLDSMHEVVENLLNYCFQTFLDKTMSIEFPEMLAEIITNQIPKYSNGNIKKLLFHQK
|
|
|
|---|
| BDBM50048341 |
|---|
| n/a |
|---|
| Name | BDBM50048341 |
|---|
| Synonyms: | CHEMBL3315063 | US9593113, Example 28 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H28N4O5 |
|---|
| Mol. Mass. | 500.5457 |
|---|
| SMILES | CC(C)([C@H]1c2ccccc2Oc2nc(ccc12)-c1ccc(cc1)C(=O)N1CCOCC1)C(=O)NC(N)=O |r| |
|---|
| Structure | 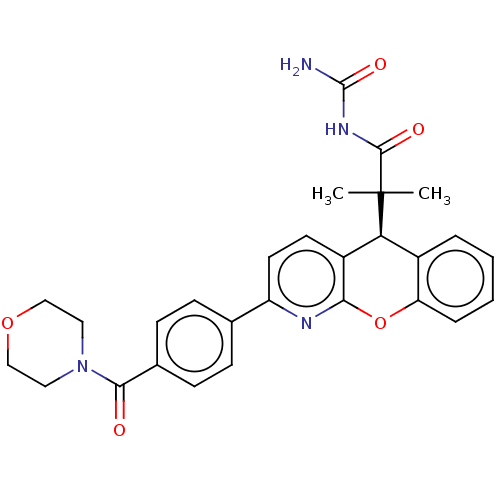
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gong, H; Yang, M; Xiao, Z; Doweyko, AM; Cunningham, M; Wang, J; Habte, S; Holloway, D; Burke, C; Shuster, D; Gao, L; Carman, J; Somerville, JE; Nadler, SG; Salter-Cid, L; Barrish, JC; Weinstein, DS Discovery of acylurea isosteres of 2-acylaminothiadiazole in the azaxanthene series of glucocorticoid receptor agonists. Bioorg Med Chem Lett24:3268-73 (2014) [PubMed] Article
Gong, H; Yang, M; Xiao, Z; Doweyko, AM; Cunningham, M; Wang, J; Habte, S; Holloway, D; Burke, C; Shuster, D; Gao, L; Carman, J; Somerville, JE; Nadler, SG; Salter-Cid, L; Barrish, JC; Weinstein, DS Discovery of acylurea isosteres of 2-acylaminothiadiazole in the azaxanthene series of glucocorticoid receptor agonists. Bioorg Med Chem Lett24:3268-73 (2014) [PubMed] Article