| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Amine oxidase [flavin-containing] B |
|---|
| Ligand | BDBM50240772 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1473015 (CHEMBL3420665) |
|---|
| IC50 | 6210±n/a nM |
|---|
| Citation |  Ma, LY; Zheng, YC; Wang, SQ; Wang, B; Wang, ZR; Pang, LP; Zhang, M; Wang, JW; Ding, L; Li, J; Wang, C; Hu, B; Liu, Y; Zhang, XD; Wang, JJ; Wang, ZJ; Zhao, W; Liu, HM Design, synthesis, and structure-activity relationship of novel LSD1 inhibitors based on pyrimidine-thiourea hybrids as potent, orally active antitumor agents. J Med Chem58:1705-16 (2015) [PubMed] Article Ma, LY; Zheng, YC; Wang, SQ; Wang, B; Wang, ZR; Pang, LP; Zhang, M; Wang, JW; Ding, L; Li, J; Wang, C; Hu, B; Liu, Y; Zhang, XD; Wang, JJ; Wang, ZJ; Zhao, W; Liu, HM Design, synthesis, and structure-activity relationship of novel LSD1 inhibitors based on pyrimidine-thiourea hybrids as potent, orally active antitumor agents. J Med Chem58:1705-16 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Amine oxidase [flavin-containing] B |
|---|
| Name: | Amine oxidase [flavin-containing] B |
|---|
| Synonyms: | AOFB_HUMAN | MAO-B | MAOB | Monoamine oxidase type B | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAOB) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 58768.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P27338 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MSNKCDVVVVGGGISGMAAAKLLHDSGLNVVVLEARDRVGGRTYTLRNQKVKYVDLGGSY
VGPTQNRILRLAKELGLETYKVNEVERLIHHVKGKSYPFRGPFPPVWNPITYLDHNNFWR
TMDDMGREIPSDAPWKAPLAEEWDNMTMKELLDKLCWTESAKQLATLFVNLCVTAETHEV
SALWFLWYVKQCGGTTRIISTTNGGQERKFVGGSGQVSERIMDLLGDRVKLERPVIYIDQ
TRENVLVETLNHEMYEAKYVISAIPPTLGMKIHFNPPLPMMRNQMITRVPLGSVIKCIVY
YKEPFWRKKDYCGTMIIDGEEAPVAYTLDDTKPEGNYAAIMGFILAHKARKLARLTKEER
LKKLCELYAKVLGSLEALEPVHYEEKNWCEEQYSGGCYTTYFPPGILTQYGRVLRQPVDR
IYFAGTETATHWSGYMEGAVEAGERAAREILHAMGKIPEDEIWQSEPESVDVPAQPITTT
FLERHLPSVPGLLRLIGLTTIFSATALGFLAHKRGLLVRV
|
|
|
|---|
| BDBM50240772 |
|---|
| n/a |
|---|
| Name | BDBM50240772 |
|---|
| Synonyms: | (1R,2S)-(-)-2-phenylcyclopropylamine | (1R,2S)-2-phenylcyclopropanamine | (tranylcypromine)2-Phenyl-cyclopropylamine | 2-Phenyl-cyclopropylamine | CHEMBL1179 | Parnate | TRANYLCYPROMINE | TRANYLCYPROMINE HYDROCHLORIDE | US10836743, Compound TCP | US8993808, Tranylcypromine | US9180183, Tranylcypromine | cid_2723716 | rel-Tranylcypromine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C9H11N |
|---|
| Mol. Mass. | 133.1903 |
|---|
| SMILES | N[C@@H]1C[C@H]1c1ccccc1 |r| |
|---|
| Structure | 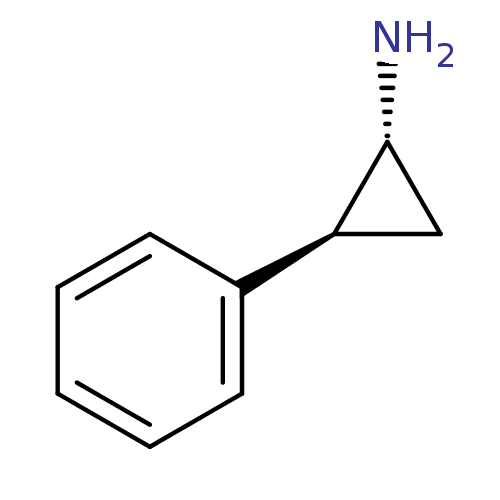
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ma, LY; Zheng, YC; Wang, SQ; Wang, B; Wang, ZR; Pang, LP; Zhang, M; Wang, JW; Ding, L; Li, J; Wang, C; Hu, B; Liu, Y; Zhang, XD; Wang, JJ; Wang, ZJ; Zhao, W; Liu, HM Design, synthesis, and structure-activity relationship of novel LSD1 inhibitors based on pyrimidine-thiourea hybrids as potent, orally active antitumor agents. J Med Chem58:1705-16 (2015) [PubMed] Article
Ma, LY; Zheng, YC; Wang, SQ; Wang, B; Wang, ZR; Pang, LP; Zhang, M; Wang, JW; Ding, L; Li, J; Wang, C; Hu, B; Liu, Y; Zhang, XD; Wang, JJ; Wang, ZJ; Zhao, W; Liu, HM Design, synthesis, and structure-activity relationship of novel LSD1 inhibitors based on pyrimidine-thiourea hybrids as potent, orally active antitumor agents. J Med Chem58:1705-16 (2015) [PubMed] Article