| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 8 |
|---|
| Ligand | BDBM15997 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1544313 (CHEMBL3751430) |
|---|
| IC50 | 28±n/a nM |
|---|
| Citation |  Simon-Szab�, L; Kokas, M; Greff, Z; Boros, S; B�nhegyi, P; Zs�kai, L; Sz�ntai-Kis, C; Vantus, T; Mandl, J; B�nhegyi, G; V�lyi-Nagy, I; Orfi, L; Ullrich, A; Csala, M; K�ri, G Novel compounds reducing IRS-1 serine phosphorylation for treatment of diabetes. Bioorg Med Chem Lett26:424-8 (2016) [PubMed] Article Simon-Szab�, L; Kokas, M; Greff, Z; Boros, S; B�nhegyi, P; Zs�kai, L; Sz�ntai-Kis, C; Vantus, T; Mandl, J; B�nhegyi, G; V�lyi-Nagy, I; Orfi, L; Ullrich, A; Csala, M; K�ri, G Novel compounds reducing IRS-1 serine phosphorylation for treatment of diabetes. Bioorg Med Chem Lett26:424-8 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 8 |
|---|
| Name: | Mitogen-activated protein kinase 8 |
|---|
| Synonyms: | JNK-46 | JNK1 | JNK1-alpha-1 | MAPK8 | MK08_HUMAN | Mitogen-Activated Protein Kinase 8 (JNK1) | PRKM8 | SAPK1 | SAPK1C | Stress-activated protein kinase JNK1 | c-Jun N-terminal kinase 1 | c-Jun N-terminal kinase 1 (JNK1) | c-Jun N-terminal kinase 1(JNK1) | c-Jun N-terminal kinase 2 (JNK2) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48297.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | JNK-1 was purchased from Upstate Cell Signaling Solutions (formerly Upstate Biotechnology). |
|---|
| Residue: | 427 |
|---|
| Sequence: | MSRSKRDNNFYSVEIGDSTFTVLKRYQNLKPIGSGAQGIVCAAYDAILERNVAIKKLSRP
FQNQTHAKRAYRELVLMKCVNHKNIIGLLNVFTPQKSLEEFQDVYIVMELMDANLCQVIQ
MELDHERMSYLLYQMLCGIKHLHSAGIIHRDLKPSNIVVKSDCTLKILDFGLARTAGTSF
MMTPYVVTRYYRAPEVILGMGYKENVDLWSVGCIMGEMVCHKILFPGRDYIDQWNKVIEQ
LGTPCPEFMKKLQPTVRTYVENRPKYAGYSFEKLFPDVLFPADSEHNKLKASQARDLLSK
MLVIDASKRISVDEALQHPYINVWYDPSEAEAPPPKIPDKQLDEREHTIEEWKELIYKEV
MDLEERTKNGVIRGQPSPLGAAVINGSQHPSSSSSVNDVSSMSTDPTLASDTDSSLEAAA
GPLGCCR
|
|
|
|---|
| BDBM15997 |
|---|
| n/a |
|---|
| Name | BDBM15997 |
|---|
| Synonyms: | 2-({2-[(3-fluorophenyl)amino]pyrimidin-4-yl}amino)benzoic acid | 4-anilinopyrimidine 8f |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H13FN4O2 |
|---|
| Mol. Mass. | 324.3091 |
|---|
| SMILES | OC(=O)c1ccccc1Nc1ccnc(Nc2cccc(F)c2)n1 |
|---|
| Structure | 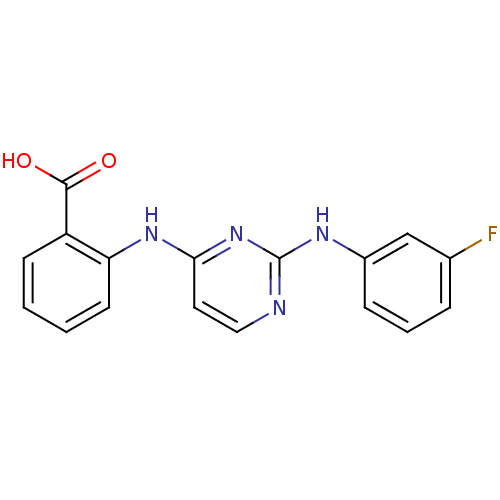
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Simon-Szab�, L; Kokas, M; Greff, Z; Boros, S; B�nhegyi, P; Zs�kai, L; Sz�ntai-Kis, C; Vantus, T; Mandl, J; B�nhegyi, G; V�lyi-Nagy, I; Orfi, L; Ullrich, A; Csala, M; K�ri, G Novel compounds reducing IRS-1 serine phosphorylation for treatment of diabetes. Bioorg Med Chem Lett26:424-8 (2016) [PubMed] Article
Simon-Szab�, L; Kokas, M; Greff, Z; Boros, S; B�nhegyi, P; Zs�kai, L; Sz�ntai-Kis, C; Vantus, T; Mandl, J; B�nhegyi, G; V�lyi-Nagy, I; Orfi, L; Ullrich, A; Csala, M; K�ri, G Novel compounds reducing IRS-1 serine phosphorylation for treatment of diabetes. Bioorg Med Chem Lett26:424-8 (2016) [PubMed] Article