| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 5 |
|---|
| Ligand | BDBM50143203 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1550897 (CHEMBL3762852) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Kusumi, K; Shinozaki, K; Yamaura, Y; Hashimoto, A; Kurata, H; Naganawa, A; Otsuki, K; Matsushita, T; Sekiguchi, T; Kakuuchi, A; Yamamoto, H; Seko, T Discovery of novel S1P2 antagonists, part 3: Improving the oral bioavailability of a series of 1,3-bis(aryloxy)benzene derivatives. Bioorg Med Chem Lett26:1209-13 (2016) [PubMed] Article Kusumi, K; Shinozaki, K; Yamaura, Y; Hashimoto, A; Kurata, H; Naganawa, A; Otsuki, K; Matsushita, T; Sekiguchi, T; Kakuuchi, A; Yamamoto, H; Seko, T Discovery of novel S1P2 antagonists, part 3: Improving the oral bioavailability of a series of 1,3-bis(aryloxy)benzene derivatives. Bioorg Med Chem Lett26:1209-13 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 5 |
|---|
| Name: | Sphingosine 1-phosphate receptor 5 |
|---|
| Synonyms: | EDG8 | Endothelial differentiation sphingolipid G-protein-coupled receptor 8 | S1P5 | S1PR5 | S1PR5_HUMAN | Sphingosine 1-phosphate receptor | Sphingosine 1-phosphate receptor Edg-8 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 41796.42 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 398 |
|---|
| Sequence: | MESGLLRPAPVSEVIVLHYNYTGKLRGARYQPGAGLRADAVVCLAVCAFIVLENLAVLLV
LGRHPRFHAPMFLLLGSLTLSDLLAGAAYAANILLSGPLTLKLSPALWFAREGGVFVALT
ASVLSLLAIALERSLTMARRGPAPVSSRGRTLAMAAAAWGVSLLLGLLPALGWNCLGRLD
ACSTVLPLYAKAYVLFCVLAFVGILAAICALYARIYCQVRANARRLPARPGTAGTTSTRA
RRKPRSLALLRTLSVVLLAFVACWGPLFLLLLLDVACPARTCPVLLQADPFLGLAMANSL
LNPIIYTLTNRDLRHALLRLVCCGRHSCGRDPSGSQQSASAAEASGGLRRCLPPGLDGSF
SGSERSSPQRDGLDTSGSTGSPGAPTAARTLVSEPAAD
|
|
|
|---|
| BDBM50143203 |
|---|
| n/a |
|---|
| Name | BDBM50143203 |
|---|
| Synonyms: | CHEMBL3759434 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H35FN2O6 |
|---|
| Mol. Mass. | 550.6178 |
|---|
| SMILES | CC(C)CC1(O)CCN(C1)C(=O)Nc1cc(Oc2ccc(F)cc2)cc(Oc2ccc(cc2)C(C)(C)C(O)=O)c1 |
|---|
| Structure | 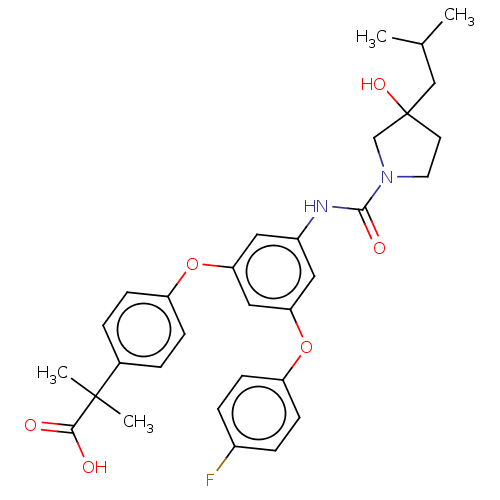
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kusumi, K; Shinozaki, K; Yamaura, Y; Hashimoto, A; Kurata, H; Naganawa, A; Otsuki, K; Matsushita, T; Sekiguchi, T; Kakuuchi, A; Yamamoto, H; Seko, T Discovery of novel S1P2 antagonists, part 3: Improving the oral bioavailability of a series of 1,3-bis(aryloxy)benzene derivatives. Bioorg Med Chem Lett26:1209-13 (2016) [PubMed] Article
Kusumi, K; Shinozaki, K; Yamaura, Y; Hashimoto, A; Kurata, H; Naganawa, A; Otsuki, K; Matsushita, T; Sekiguchi, T; Kakuuchi, A; Yamamoto, H; Seko, T Discovery of novel S1P2 antagonists, part 3: Improving the oral bioavailability of a series of 1,3-bis(aryloxy)benzene derivatives. Bioorg Med Chem Lett26:1209-13 (2016) [PubMed] Article