| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50180840 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1584799 (CHEMBL3821882) |
|---|
| IC50 | 19100±n/a nM |
|---|
| Citation |  Carosati, E; Cosimelli, B; Ioan, P; Severi, E; Katneni, K; Chiu, FC; Saponara, S; Fusi, F; Frosini, M; Matucci, R; Micucci, M; Chiarini, A; Spinelli, D; Budriesi, R Understanding Oxadiazolothiazinone Biological Properties: Negative Inotropic Activity versus Cytochrome P450-Mediated Metabolism. J Med Chem59:3340-52 (2016) [PubMed] Article Carosati, E; Cosimelli, B; Ioan, P; Severi, E; Katneni, K; Chiu, FC; Saponara, S; Fusi, F; Frosini, M; Matucci, R; Micucci, M; Chiarini, A; Spinelli, D; Budriesi, R Understanding Oxadiazolothiazinone Biological Properties: Negative Inotropic Activity versus Cytochrome P450-Mediated Metabolism. J Med Chem59:3340-52 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50180840 |
|---|
| n/a |
|---|
| Name | BDBM50180840 |
|---|
| Synonyms: | CHEMBL3819185 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H14N2O3S |
|---|
| Mol. Mass. | 290.338 |
|---|
| SMILES | CCOC1(SC=C(C)n2c1noc2=O)c1ccccc1 |t:5| |
|---|
| Structure | 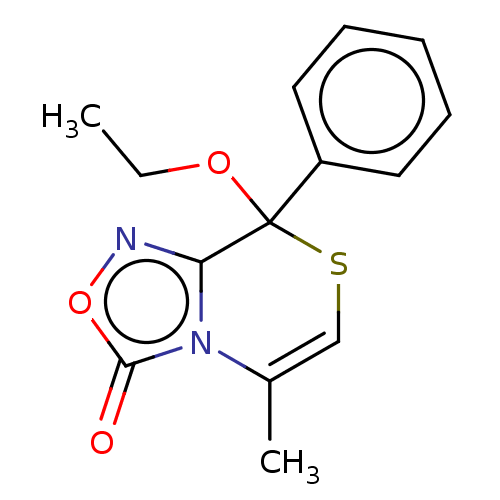
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Carosati, E; Cosimelli, B; Ioan, P; Severi, E; Katneni, K; Chiu, FC; Saponara, S; Fusi, F; Frosini, M; Matucci, R; Micucci, M; Chiarini, A; Spinelli, D; Budriesi, R Understanding Oxadiazolothiazinone Biological Properties: Negative Inotropic Activity versus Cytochrome P450-Mediated Metabolism. J Med Chem59:3340-52 (2016) [PubMed] Article
Carosati, E; Cosimelli, B; Ioan, P; Severi, E; Katneni, K; Chiu, FC; Saponara, S; Fusi, F; Frosini, M; Matucci, R; Micucci, M; Chiarini, A; Spinelli, D; Budriesi, R Understanding Oxadiazolothiazinone Biological Properties: Negative Inotropic Activity versus Cytochrome P450-Mediated Metabolism. J Med Chem59:3340-52 (2016) [PubMed] Article