| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Aryl hydrocarbon receptor |
|---|
| Ligand | BDBM50185305 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1587673 (CHEMBL3825787) |
|---|
| EC50 | 2300±n/a nM |
|---|
| Citation |  Smutny, T; Nova, A; Drechslerov�, M; Carazo, A; Hyrsova, L; Hru?kov�, ZR; Kune?, J; Pour, M; ?pul�k, M; Pavek, P 2-(3-Methoxyphenyl)quinazoline Derivatives: A New Class of Direct Constitutive Androstane Receptor (CAR) Agonists. J Med Chem59:4601-10 (2016) [PubMed] Article Smutny, T; Nova, A; Drechslerov�, M; Carazo, A; Hyrsova, L; Hru?kov�, ZR; Kune?, J; Pour, M; ?pul�k, M; Pavek, P 2-(3-Methoxyphenyl)quinazoline Derivatives: A New Class of Direct Constitutive Androstane Receptor (CAR) Agonists. J Med Chem59:4601-10 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Aryl hydrocarbon receptor |
|---|
| Name: | Aryl hydrocarbon receptor |
|---|
| Synonyms: | AHR | AHR_HUMAN | BHLHE76 | Class E basic helix-loop-helix protein 76 | aryl hydrocarbon receptor precursor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 96143.77 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1503828 |
|---|
| Residue: | 848 |
|---|
| Sequence: | MNSSSANITYASRKRRKPVQKTVKPIPAEGIKSNPSKRHRDRLNTELDRLASLLPFPQDV
INKLDKLSVLRLSVSYLRAKSFFDVALKSSPTERNGGQDNCRAANFREGLNLQEGEFLLQ
ALNGFVLVVTTDALVFYASSTIQDYLGFQQSDVIHQSVYELIHTEDRAEFQRQLHWALNP
SQCTESGQGIEEATGLPQTVVCYNPDQIPPENSPLMERCFICRLRCLLDNSSGFLAMNFQ
GKLKYLHGQKKKGKDGSILPPQLALFAIATPLQPPSILEIRTKNFIFRTKHKLDFTPIGC
DAKGRIVLGYTEAELCTRGSGYQFIHAADMLYCAESHIRMIKTGESGMIVFRLLTKNNRW
TWVQSNARLLYKNGRPDYIIVTQRPLTDEEGTEHLRKRNTKLPFMFTTGEAVLYEATNPF
PAIMDPLPLRTKNGTSGKDSATTSTLSKDSLNPSSLLAAMMQQDESIYLYPASSTSSTAP
FENNFFNESMNECRNWQDNTAPMGNDTILKHEQIDQPQDVNSFAGGHPGLFQDSKNSDLY
SIMKNLGIDFEDIRHMQNEKFFRNDFSGEVDFRDIDLTDEILTYVQDSLSKSPFIPSDYQ
QQQSLALNSSCMVQEHLHLEQQQQHHQKQVVVEPQQQLCQKMKHMQVNGMFENWNSNQFV
PFNCPQQDPQQYNVFTDLHGISQEFPYKSEMDSMPYTQNFISCNQPVLPQHSKCTELDYP
MGSFEPSPYPTTSSLEDFVTCLQLPENQKHGLNPQSAIITPQTCYAGAVSMYQCQPEPQH
THVGQMQYNPVLPGQQAFLNKFQNGVLNETYPAELNNINNTQTTTHLQPLHHPSEARPFP
DLTSSGFL
|
|
|
|---|
| BDBM50185305 |
|---|
| n/a |
|---|
| Name | BDBM50185305 |
|---|
| Synonyms: | CHEMBL3822867 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H14N2OS |
|---|
| Mol. Mass. | 282.36 |
|---|
| SMILES | COc1cccc(c1)-c1nc(SC)c2ccccc2n1 |
|---|
| Structure | 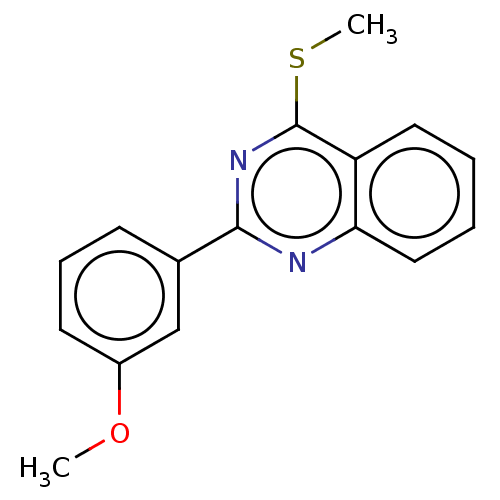
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Smutny, T; Nova, A; Drechslerov�, M; Carazo, A; Hyrsova, L; Hru?kov�, ZR; Kune?, J; Pour, M; ?pul�k, M; Pavek, P 2-(3-Methoxyphenyl)quinazoline Derivatives: A New Class of Direct Constitutive Androstane Receptor (CAR) Agonists. J Med Chem59:4601-10 (2016) [PubMed] Article
Smutny, T; Nova, A; Drechslerov�, M; Carazo, A; Hyrsova, L; Hru?kov�, ZR; Kune?, J; Pour, M; ?pul�k, M; Pavek, P 2-(3-Methoxyphenyl)quinazoline Derivatives: A New Class of Direct Constitutive Androstane Receptor (CAR) Agonists. J Med Chem59:4601-10 (2016) [PubMed] Article