| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Hematopoietic prostaglandin D synthase |
|---|
| Ligand | BDBM50240648 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | >10000±n/a nM |
|---|
| Comments | PDSP_4303 |
|---|
| Citation |  Abramovitz, M; Adam, M; Boie, Y; Carrière, M; Denis, D; Godbout, C; Lamontagne, S; Rochette, C; Sawyer, N; Tremblay, NM; Belley, M; Gallant, M; Dufresne, C; Gareau, Y; Ruel, R; Juteau, H; Labelle, M; Ouimet, N; Metters, KM The utilization of recombinant prostanoid receptors to determine the affinities and selectivities of prostaglandins and related analogs. Biochim Biophys Acta1483:285-93 (2000) [PubMed] Article Abramovitz, M; Adam, M; Boie, Y; Carrière, M; Denis, D; Godbout, C; Lamontagne, S; Rochette, C; Sawyer, N; Tremblay, NM; Belley, M; Gallant, M; Dufresne, C; Gareau, Y; Ruel, R; Juteau, H; Labelle, M; Ouimet, N; Metters, KM The utilization of recombinant prostanoid receptors to determine the affinities and selectivities of prostaglandins and related analogs. Biochim Biophys Acta1483:285-93 (2000) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Hematopoietic prostaglandin D synthase |
|---|
| Name: | Hematopoietic prostaglandin D synthase |
|---|
| Synonyms: | GSTS | Glutathione-dependent PGD synthetase | Glutathione-requiring prostaglandin D synthase | H-PGDS | HPGDS | HPGDS_HUMAN | Hematopoietic prostaglandin D synthase | Hematopoietic prostaglandin D synthase (H-PGDS) | Hematopoietic prostaglandin D synthase (HPGDS) | PGDS | PTGDS2 | Prostaglandin D | Prostaglandin D Synthase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 23341.07 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The protein was expressed in E. coli strain BL21(DE3) with an N-terminal 6-His tag. |
|---|
| Residue: | 199 |
|---|
| Sequence: | MPNYKLTYFNMRGRAEIIRYIFAYLDIQYEDHRIEQADWPEIKSTLPFGKIPILEVDGLT
LHQSLAIARYLTKNTDLAGNTEMEQCHVDAIVDTLDDFMSCFPWAEKKQDVKEQMFNELL
TYNAPHLMQDLDTYLGGREWLIGNSVTWADFYWEICSTTLLVFKPDLLDNHPRLVTLRKK
VQAIPAVANWIKRRPQTKL
|
|
|
|---|
| BDBM50240648 |
|---|
| n/a |
|---|
| Name | BDBM50240648 |
|---|
| Synonyms: | LATANOPROST (FREE ACID) | PhXA 41 | Xalatan | isopropyl (5Z,9alpha,11alpha,15R)-9,11,15-trihydroxy-17-phenyl-18,19,20-trinorprost-5-en-1-oate | isopropyl (Z)-7-((1R,2R,3R,5S)-3,5-dihydroxy-2-((3R)-3-hydroxy-5-phenylpentyl)cyclopentyl)-5-heptenoate | latanoprost | propan-2-yl (5Z)-7-{(1R,2R,3R,5S)-3,5-dihydroxy-2-[(3R)-3-hydroxy-5-phenylpentyl]cyclopentyl}hept-5-enoate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H40O5 |
|---|
| Mol. Mass. | 432.5928 |
|---|
| SMILES | CC(C)OC(=O)CCC\C=C/C[C@H]1[C@@H](O)C[C@@H](O)[C@@H]1CC[C@@H](O)CCc1ccccc1 |r| |
|---|
| Structure | 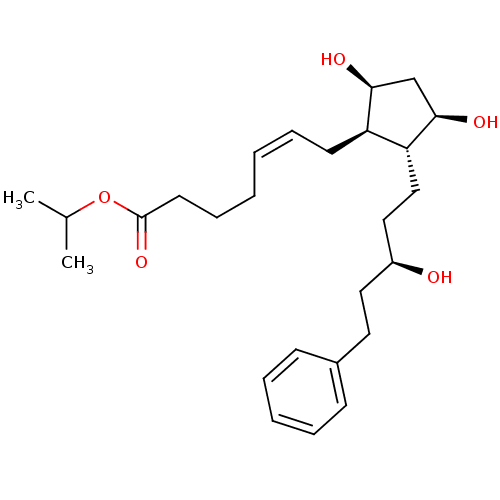
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Abramovitz, M; Adam, M; Boie, Y; Carrière, M; Denis, D; Godbout, C; Lamontagne, S; Rochette, C; Sawyer, N; Tremblay, NM; Belley, M; Gallant, M; Dufresne, C; Gareau, Y; Ruel, R; Juteau, H; Labelle, M; Ouimet, N; Metters, KM The utilization of recombinant prostanoid receptors to determine the affinities and selectivities of prostaglandins and related analogs. Biochim Biophys Acta1483:285-93 (2000) [PubMed] Article
Abramovitz, M; Adam, M; Boie, Y; Carrière, M; Denis, D; Godbout, C; Lamontagne, S; Rochette, C; Sawyer, N; Tremblay, NM; Belley, M; Gallant, M; Dufresne, C; Gareau, Y; Ruel, R; Juteau, H; Labelle, M; Ouimet, N; Metters, KM The utilization of recombinant prostanoid receptors to determine the affinities and selectivities of prostaglandins and related analogs. Biochim Biophys Acta1483:285-93 (2000) [PubMed] Article