| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1B |
|---|
| Ligand | BDBM50050467 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 1000±n/a nM |
|---|
| Comments | PDSP_3305 |
|---|
| Citation |  Millan, MJ; Gobert, A; Newman-Tancredi, A; Lejeune, F; Cussac, D; Rivet, JM; Audinot, V; Dubuffet, T; Lavielle, G S33084, a novel, potent, selective, and competitive antagonist at dopamine D(3)-receptors: I. Receptorial, electrophysiological and neurochemical profile compared with GR218,231 and L741,626. J Pharmacol Exp Ther293:1048-62 (2000) [PubMed] Millan, MJ; Gobert, A; Newman-Tancredi, A; Lejeune, F; Cussac, D; Rivet, JM; Audinot, V; Dubuffet, T; Lavielle, G S33084, a novel, potent, selective, and competitive antagonist at dopamine D(3)-receptors: I. Receptorial, electrophysiological and neurochemical profile compared with GR218,231 and L741,626. J Pharmacol Exp Ther293:1048-62 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| 5-hydroxytryptamine receptor 1B |
|---|
| Name: | 5-hydroxytryptamine receptor 1B |
|---|
| Synonyms: | 5-HT-1B | 5-HT-1D-beta | 5-HT1B | 5-hydroxytryptamine receptor 1B (5-HT1B) | 5HT1B_HUMAN | HTR1B | HTR1DB | S12 | Serotonin (5-HT) receptor | Serotonin 1D beta receptor | Serotonin Receptor 1B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 43579.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells |
|---|
| Residue: | 390 |
|---|
| Sequence: | MEEPGAQCAPPPPAGSETWVPQANLSSAPSQNCSAKDYIYQDSISLPWKVLLVMLLALIT
LATTLSNAFVIATVYRTRKLHTPANYLIASLAVTDLLVSILVMPISTMYTVTGRWTLGQV
VCDFWLSSDITCCTASILHLCVIALDRYWAITDAVEYSAKRTPKRAAVMIALVWVFSISI
SLPPFFWRQAKAEEEVSECVVNTDHILYTVYSTVGAFYFPTLLLIALYGRIYVEARSRIL
KQTPNRTGKRLTRAQLITDSPGSTSSVTSINSRVPDVPSESGSPVYVNQVKVRVSDALLE
KKKLMAARERKATKTLGIILGAFIVCWLPFFIISLVMPICKDACWFHLAIFDFFTWLGYL
NSLINPIIYTMSNEDFKQAFHKLIRFKCTS
|
|
|
|---|
| BDBM50050467 |
|---|
| n/a |
|---|
| Name | BDBM50050467 |
|---|
| Synonyms: | 1-((1H-indol-3-yl)methyl)-4-(4-chlorophenyl)piperidin-4-ol | 3-[4-(4-chlorophenyl)-4-hydroxypiperidinyl]methylindole | 4-(4-Chloro-phenyl)-1-(1H-indol-3-ylmethyl)-piperidin-4-ol | CHEMBL445102 | L-741626 | L741,626 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H21ClN2O |
|---|
| Mol. Mass. | 340.847 |
|---|
| SMILES | OC1(CCN(Cc2c[nH]c3ccccc23)CC1)c1ccc(Cl)cc1 |
|---|
| Structure | 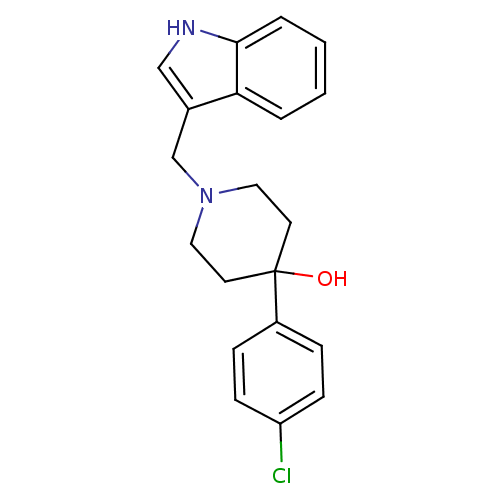
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Millan, MJ; Gobert, A; Newman-Tancredi, A; Lejeune, F; Cussac, D; Rivet, JM; Audinot, V; Dubuffet, T; Lavielle, G S33084, a novel, potent, selective, and competitive antagonist at dopamine D(3)-receptors: I. Receptorial, electrophysiological and neurochemical profile compared with GR218,231 and L741,626. J Pharmacol Exp Ther293:1048-62 (2000) [PubMed]
Millan, MJ; Gobert, A; Newman-Tancredi, A; Lejeune, F; Cussac, D; Rivet, JM; Audinot, V; Dubuffet, T; Lavielle, G S33084, a novel, potent, selective, and competitive antagonist at dopamine D(3)-receptors: I. Receptorial, electrophysiological and neurochemical profile compared with GR218,231 and L741,626. J Pharmacol Exp Ther293:1048-62 (2000) [PubMed]