| Reaction Details |
|---|
 | Report a problem with these data |
| Target | HIV-1 protease |
|---|
| Ligand | BDBM93238 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Fluorometric Assay |
|---|
| Ki | 118±0.0 nM |
|---|
| Citation |  Wannberg, J; Kaiser, NF; Vrang, L; Samuelsson, B; Larhed, M; Hallberg, A High-speed synthesis of potent C2-symmetric HIV-1 protease inhibitors by in-situ aminocarbonylations. J Comb Chem7:611-7 (2005) [PubMed] Article Wannberg, J; Kaiser, NF; Vrang, L; Samuelsson, B; Larhed, M; Hallberg, A High-speed synthesis of potent C2-symmetric HIV-1 protease inhibitors by in-situ aminocarbonylations. J Comb Chem7:611-7 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| HIV-1 protease |
|---|
| Name: | HIV-1 protease |
|---|
| Synonyms: | n/a |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 10794.16 |
|---|
| Organism: | Human immunodeficiency virus |
|---|
| Description: | Q9YQ34 |
|---|
| Residue: | 99 |
|---|
| Sequence: | PQITLWQRPLVTVKIGGQLKEALLDTGADDTVLEEMNLPGRWKPKMIGGIGGFIKVRQYD
QILVEICGHKAIGTVLVGPTPVNIIGRNLLTQIGCTLNF
|
|
|
|---|
| BDBM93238 |
|---|
| n/a |
|---|
| Name | BDBM93238 |
|---|
| Synonyms: | HIV-1 Protease Inhibitor, 6i |
|---|
| Type | Small molecule |
|---|
| Emp. Form. | C44H54N8O10 |
|---|
| Mol. Mass. | 854.9472 |
|---|
| SMILES | CNC(=O)[C@@H](NC(=O)[C@H](OCc1ccccc1C(=O)Nc1cccnc1)[C@H](O)[C@@H](O)[C@@H](OCc1ccccc1C(=O)Nc1cccnc1)C(=O)N[C@@H](C(C)C)C(=O)NC)C(C)C |r| |
|---|
| Structure | 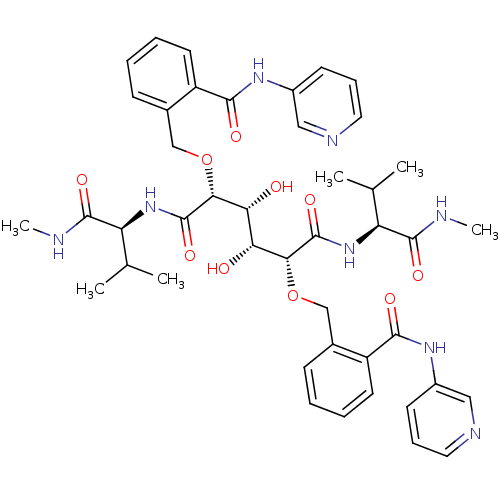
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wannberg, J; Kaiser, NF; Vrang, L; Samuelsson, B; Larhed, M; Hallberg, A High-speed synthesis of potent C2-symmetric HIV-1 protease inhibitors by in-situ aminocarbonylations. J Comb Chem7:611-7 (2005) [PubMed] Article
Wannberg, J; Kaiser, NF; Vrang, L; Samuelsson, B; Larhed, M; Hallberg, A High-speed synthesis of potent C2-symmetric HIV-1 protease inhibitors by in-situ aminocarbonylations. J Comb Chem7:611-7 (2005) [PubMed] Article