| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Macrophage-stimulating protein receptor |
|---|
| Ligand | BDBM50399540 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | HTRF kinase assay |
|---|
| pH | 7±n/a |
|---|
| Temperature | 298.15±n/a K |
|---|
| IC50 | 3.52±0.0 nM |
|---|
| Comments | extracted |
|---|
| Citation |  Zhou, S; Ren, J; Liu, M; Ren, L; Liu, Y; Gong, P Design, synthesis and pharmacological evaluation of 6,7-disubstituted-4-phenoxyquinoline derivatives as potential antitumor agents. Bioorg Chem57:30-42 (2014) [PubMed] Article Zhou, S; Ren, J; Liu, M; Ren, L; Liu, Y; Gong, P Design, synthesis and pharmacological evaluation of 6,7-disubstituted-4-phenoxyquinoline derivatives as potential antitumor agents. Bioorg Chem57:30-42 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Macrophage-stimulating protein receptor |
|---|
| Name: | Macrophage-stimulating protein receptor |
|---|
| Synonyms: | 2.7.10.1 | CD_antigen=CD136 | CDw136 | MSP receptor | MST1R | Macrophage-stimulating protein receptor (MST1R) | Macrophage-stimulating protein receptor alpha chain | Macrophage-stimulating protein receptor beta chain | PTK8 | Protein-tyrosine kinase 8 | RON | RON_HUMAN | Tyrosine kinase receptor ron | p185-Ron |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 152270.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q04912 |
|---|
| Residue: | 1400 |
|---|
| Sequence: | MELLPPLPQSFLLLLLLPAKPAAGEDWQCPRTPYAASRDFDVKYVVPSFSAGGLVQAMVT
YEGDRNESAVFVAIRNRLHVLGPDLKSVQSLATGPAGDPGCQTCAACGPGPHGPPGDTDT
KVLVLDPALPALVSCGSSLQGRCFLHDLEPQGTAVHLAAPACLFSAHHNRPDDCPDCVAS
PLGTRVTVVEQGQASYFYVASSLDAAVAASFSPRSVSIRRLKADASGFAPGFVALSVLPK
HLVSYSIEYVHSFHTGAFVYFLTVQPASVTDDPSALHTRLARLSATEPELGDYRELVLDC
RFAPKRRRRGAPEGGQPYPVLRVAHSAPVGAQLATELSIAEGQEVLFGVFVTGKDGGPGV
GPNSVVCAFPIDLLDTLIDEGVERCCESPVHPGLRRGLDFFQSPSFCPNPPGLEALSPNT
SCRHFPLLVSSSFSRVDLFNGLLGPVQVTALYVTRLDNVTVAHMGTMDGRILQVELVRSL
NYLLYVSNFSLGDSGQPVQRDVSRLGDHLLFASGDQVFQVPIQGPGCRHFLTCGRCLRAW
HFMGCGWCGNMCGQQKECPGSWQQDHCPPKLTEFHPHSGPLRGSTRLTLCGSNFYLHPSG
LVPEGTHQVTVGQSPCRPLPKDSSKLRPVPRKDFVEEFECELEPLGTQAVGPTNVSLTVT
NMPPGKHFRVDGTSVLRGFSFMEPVLIAVQPLFGPRAGGTCLTLEGQSLSVGTSRAVLVN
GTECLLARVSEGQLLCATPPGATVASVPLSLQVGGAQVPGSWTFQYREDPVVLSISPNCG
YINSHITICGQHLTSAWHLVLSFHDGLRAVESRCERQLPEQQLCRLPEYVVRDPQGWVAG
NLSARGDGAAGFTLPGFRFLPPPHPPSANLVPLKPEEHAIKFEYIGLGAVADCVGINVTV
GGESCQHEFRGDMVVCPLPPSLQLGQDGAPLQVCVDGECHILGRVVRPGPDGVPQSTLLG
ILLPLLLLVAALATALVFSYWWRRKQLVLPPNLNDLASLDQTAGATPLPILYSGSDYRSG
LALPAIDGLDSTTCVHGASFSDSEDESCVPLLRKESIQLRDLDSALLAEVKDVLIPHERV
VTHSDRVIGKGHFGVVYHGEYIDQAQNRIQCAIKSLSRITEMQQVEAFLREGLLMRGLNH
PNVLALIGIMLPPEGLPHVLLPYMCHGDLLQFIRSPQRNPTVKDLISFGLQVARGMEYLA
EQKFVHRDLAARNCMLDESFTVKVADFGLARDILDREYYSVQQHRHARLPVKWMALESLQ
TYRFTTKSDVWSFGVLLWELLTRGAPPYRHIDPFDLTHFLAQGRRLPQPEYCPDSLYQVM
QQCWEADPAVRPTFRVLVGEVEQIVSALLGDHYVQLPATYMNLGPSTSHEMNVRPEQPQF
SPMPGNVRRPRPLSEPPRPT
|
|
|
|---|
| BDBM50399540 |
|---|
| n/a |
|---|
| Name | BDBM50399540 |
|---|
| Synonyms: | FORETINIB | US10464902, Foretinib | US10882853, Compound For-Oxide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C34H34F2N4O6 |
|---|
| Mol. Mass. | 632.6538 |
|---|
| SMILES | COc1cc2c(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)ccnc2cc1OCCCN1CCOCC1 |
|---|
| Structure | 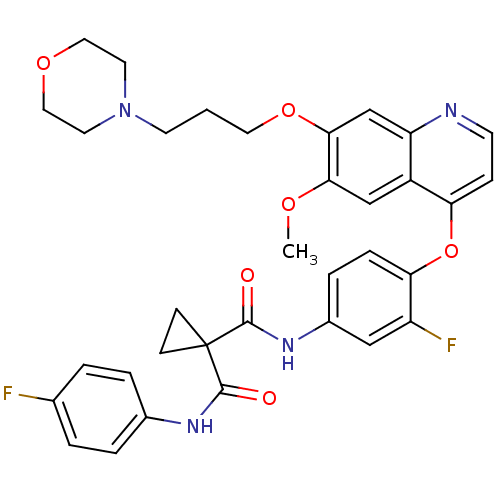
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhou, S; Ren, J; Liu, M; Ren, L; Liu, Y; Gong, P Design, synthesis and pharmacological evaluation of 6,7-disubstituted-4-phenoxyquinoline derivatives as potential antitumor agents. Bioorg Chem57:30-42 (2014) [PubMed] Article
Zhou, S; Ren, J; Liu, M; Ren, L; Liu, Y; Gong, P Design, synthesis and pharmacological evaluation of 6,7-disubstituted-4-phenoxyquinoline derivatives as potential antitumor agents. Bioorg Chem57:30-42 (2014) [PubMed] Article