

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Protein Nef | ||
| Ligand | BDBM288115 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | Measurement of Antiviral (HIV-1) Activity | ||
| Temperature | 298.15±n/a K | ||
| EC50 | 0.900±n/a nM | ||
| Comments | extracted | ||
| Citation |  Miyazaki, S; Isoshima, H; Oshita, K; Kawashita, S; Nagahashi, N; Terashita, M Substituted spiropyrido[1,2-a]pyrazine derivative and medicinal use thereof as HIV integrase inhibitor US Patent US10087178 Publication Date 10/2/2018 Miyazaki, S; Isoshima, H; Oshita, K; Kawashita, S; Nagahashi, N; Terashita, M Substituted spiropyrido[1,2-a]pyrazine derivative and medicinal use thereof as HIV integrase inhibitor US Patent US10087178 Publication Date 10/2/2018 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Protein Nef | |||
| Name: | Protein Nef | ||
| Synonyms: | 3ORF | F-protein | HIV-1 Nef | NEF_HV1BR | Negative factor | Protein Nef | nef | ||
| Type: | C-terminal core protein | ||
| Mol. Mass.: | 23340.47 | ||
| Organism: | Human immunodeficiency virus type 1 (isolate BRU/LAI group M subtype B) | ||
| Description: | P03406 | ||
| Residue: | 206 | ||
| Sequence: |
| ||
| BDBM288115 | |||
| n/a | |||
| Name | BDBM288115 | ||
| Synonyms: | Production of N-(3-chloro-2-fluorobenzyl)-9′-hydroxy-cis-3-(methoxymethyl)-2′-methyl-1′,8′-dioxo-1′,2′,3′,8′-tetrahydrospiro[cyclobutane-1,4′-pyrido[1,2-a]pyrazine]-7′-carboxamide 4-methylbenzenesulfonate | US10087178, Example 33 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C22H23ClFN3O5 | ||
| Mol. Mass. | 463.887 | ||
| SMILES | COC[C@H]1C[C@@]2(C1)CN(C)C(=O)c1c(O)c(=O)c(cn21)C(=O)NCc1cccc(Cl)c1F |r,wU:5.5,wD:3.2,(10.06,-13.22,;8.72,-13.99,;7.39,-13.22,;7.39,-11.68,;8.48,-10.59,;7.39,-9.5,;6.3,-10.59,;8.72,-8.73,;8.72,-7.19,;10.06,-6.42,;7.39,-6.42,;7.39,-4.88,;6.05,-7.19,;4.72,-6.42,;4.72,-4.88,;3.39,-7.19,;2.05,-6.42,;3.39,-8.73,;4.72,-9.5,;6.05,-8.73,;2.05,-9.5,;2.05,-11.04,;.72,-8.73,;-.61,-9.5,;-1.95,-8.73,;-1.95,-7.19,;-3.28,-6.42,;-4.61,-7.19,;-4.61,-8.73,;-5.95,-9.5,;-3.28,-9.5,;-3.28,-11.04,)| | ||
| Structure | 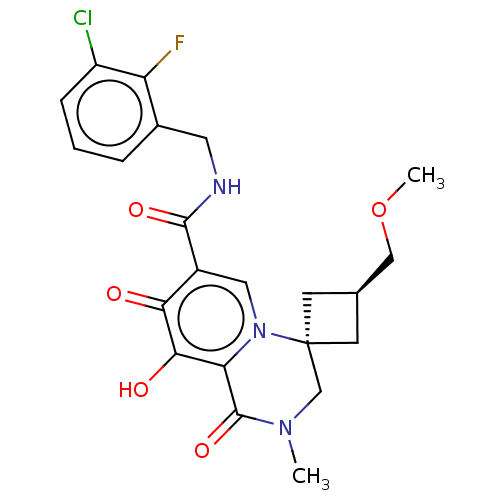
| ||