| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Serum paraoxonase/arylesterase 1 |
|---|
| Ligand | BDBM50335522 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | PON Activity Assay |
|---|
| Ki | 1.8300e+7± 1.3000e+7 nM |
|---|
| Citation |  Dilek, EB; K�frevioglu, ÿİ; Beydemir, ſ Impacts of some antibiotics on human serum paraoxonase 1 activity. J Enzyme Inhib Med Chem28:758-64 (2013) [PubMed] Article Dilek, EB; K�frevioglu, ÿİ; Beydemir, ſ Impacts of some antibiotics on human serum paraoxonase 1 activity. J Enzyme Inhib Med Chem28:758-64 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Serum paraoxonase/arylesterase 1 |
|---|
| Name: | Serum paraoxonase/arylesterase 1 |
|---|
| Synonyms: | A-esterase 1 | Aromatic esterase 1 | K-45 | PON | PON 1 | PON1 | PON1_HUMAN | Paraoxonase (PON1) | Paraoxonase (hPON1) | Paraoxonase 1 (PON1) | Serum aryldialkylphosphatase 1 | Serum paraoxonase 1 (PON1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 39719.73 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P27169 |
|---|
| Residue: | 355 |
|---|
| Sequence: | MAKLIALTLLGMGLALFRNHQSSYQTRLNALREVQPVELPNCNLVKGIETGSEDLEILPN
GLAFISSGLKYPGIKSFNPNSPGKILLMDLNEEDPTVLELGITGSKFDVSSFNPHGISTF
TDEDNAMYLLVVNHPDAKSTVELFKFQEEEKSLLHLKTIRHKLLPNLNDIVAVGPEHFYG
TNDHYFLDPYLQSWEMYLGLAWSYVVYYSPSEVRVVAEGFDFANGINISPDGKYVYIAEL
LAHKIHVYEKHANWTLTPLKSLDFNTLVDNISVDPETGDLWVGCHPNGMKIFFYDSENPP
ASEVLRIQNILTEEPKVTQVYAENGTVLQGSTVASVYKGKLLIGTVFHKALYCEL
|
|
|
|---|
| BDBM50335522 |
|---|
| n/a |
|---|
| Name | BDBM50335522 |
|---|
| Synonyms: | (2S,4R)-N-((1R,2R)-2-hydroxy-1-((2R,3R,4S,5R,6R)-3,4,5-trihydroxy-6-(methylthio)-tetrahydro-2H-pyran-2-yl)propyl)-1-methyl-4-propylpyrrolidine-2-carboxamide | (2S,4R)-N-((1S)-2-hydroxy-1-((2S,3R,4S,5R,6R)-3,4,5-trihydroxy-6-(methylthio)-tetrahydro-2H-pyran-2-yl)propyl)-1-methyl-4-propylpyrrolidine-2-carboxamide | 1-Methyl-4-propyl-pyrrolidine-2-carboxylic acid [2-hydroxy-1-(3,4,5-trihydroxy-6-methylsulfanyl-tetrahydro-pyran-2-yl)-propyl]-amide | CHEMBL1447 | Cillimycin | Frademicina | LINCOMYCIN | Lincocin | Lincomycin hydrochloride | lincomycin A |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H34N2O6S |
|---|
| Mol. Mass. | 406.537 |
|---|
| SMILES | CCC[C@@H]1C[C@H](N(C)C1)C(=O)N[C@H]([C@@H](C)O)[C@H]1O[C@H](SC)[C@H](O)[C@@H](O)[C@H]1O |r| |
|---|
| Structure | 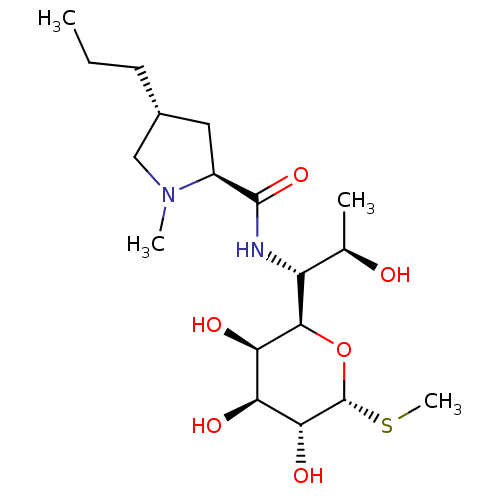
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dilek, EB; K�frevioglu, ÿİ; Beydemir, ſ Impacts of some antibiotics on human serum paraoxonase 1 activity. J Enzyme Inhib Med Chem28:758-64 (2013) [PubMed] Article
Dilek, EB; K�frevioglu, ÿİ; Beydemir, ſ Impacts of some antibiotics on human serum paraoxonase 1 activity. J Enzyme Inhib Med Chem28:758-64 (2013) [PubMed] Article