| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Ligand | BDBM340091 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Enzyme Activity Assay |
|---|
| EC50 | >1000±n/a nM |
|---|
| Citation |  Barf, TA; Jans, CG; de Man, AP; Oubrie, AA; Raaijmakers, HC; Rewinkel, JB; Sterrenburg, JG; Wijkmans, JC 4-imidazopyridazin-1-yl-benzamides as Btk inhibitors US Patent US10934296 Publication Date 3/2/2021 Barf, TA; Jans, CG; de Man, AP; Oubrie, AA; Raaijmakers, HC; Rewinkel, JB; Sterrenburg, JG; Wijkmans, JC 4-imidazopyridazin-1-yl-benzamides as Btk inhibitors US Patent US10934296 Publication Date 3/2/2021 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Proto-oncogene tyrosine-protein kinase Src |
|---|
| Name: | Proto-oncogene tyrosine-protein kinase Src |
|---|
| Synonyms: | Calmodulin/Proto-oncogene tyrosine-protein kinase Src | Protein cereblon/Tyrosine-protein kinase SRC | Proto-oncogene c-Src | Proto-oncogene tyrosine-protein kinase Src (c-Src) | SRC | SRC1 | SRC_HUMAN | Tyrosine-protein kinase Src (SRC) | V-src sarcoma (Schmidt-Ruppin A-2) viral oncogene homolog (avian) | c-Src | p60-Src | pp60c-src |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 59838.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P12931 |
|---|
| Residue: | 536 |
|---|
| Sequence: | MGSNKSKPKDASQRRRSLEPAENVHGAGGGAFPASQTPSKPASADGHRGPSAAFAPAAAE
PKLFGGFNSSDTVTSPQRAGPLAGGVTTFVALYDYESRTETDLSFKKGERLQIVNNTEGD
WWLAHSLSTGQTGYIPSNYVAPSDSIQAEEWYFGKITRRESERLLLNAENPRGTFLVRES
ETTKGAYCLSVSDFDNAKGLNVKHYKIRKLDSGGFYITSRTQFNSLQQLVAYYSKHADGL
CHRLTTVCPTSKPQTQGLAKDAWEIPRESLRLEVKLGQGCFGEVWMGTWNGTTRVAIKTL
KPGTMSPEAFLQEAQVMKKLRHEKLVQLYAVVSEEPIYIVTEYMSKGSLLDFLKGETGKY
LRLPQLVDMAAQIASGMAYVERMNYVHRDLRAANILVGENLVCKVADFGLARLIEDNEYT
ARQGAKFPIKWTAPEAALYGRFTIKSDVWSFGILLTELTTKGRVPYPGMVNREVLDQVER
GYRMPCPPECPESLHDLMCQCWRKEPEERPTFEYLQAFLEDYFTSTEPQYQPGENL
|
|
|
|---|
| BDBM340091 |
|---|
| n/a |
|---|
| Name | BDBM340091 |
|---|
| Synonyms: | 4-(8-amino-3-((S)-1-(2- chloropyrimidine-4- carbonyl)piperidin-2-yl)imidazo[1,5- a]pyrazin-1-yl)-3-methyl-N-(pyridin- 2-yl)benzamide | US10239883, Example 56 | US10934296, Example 56 | US9758524, Example 56 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H26ClN9O2 |
|---|
| Mol. Mass. | 568.029 |
|---|
| SMILES | Cc1cc(ccc1-c1nc([C@@H]2CCCCN2C(=O)c2ccnc(Cl)n2)n2ccnc(N)c12)C(=O)Nc1ccccn1 |r| |
|---|
| Structure | 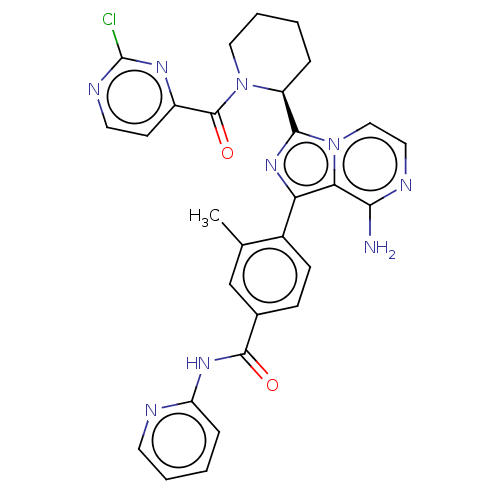
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Barf, TA; Jans, CG; de Man, AP; Oubrie, AA; Raaijmakers, HC; Rewinkel, JB; Sterrenburg, JG; Wijkmans, JC 4-imidazopyridazin-1-yl-benzamides as Btk inhibitors US Patent US10934296 Publication Date 3/2/2021
Barf, TA; Jans, CG; de Man, AP; Oubrie, AA; Raaijmakers, HC; Rewinkel, JB; Sterrenburg, JG; Wijkmans, JC 4-imidazopyridazin-1-yl-benzamides as Btk inhibitors US Patent US10934296 Publication Date 3/2/2021