null
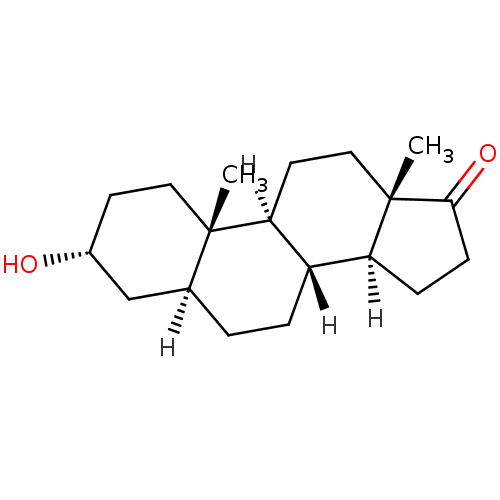
SMILES [H][C@@]12CCC(=O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])C[C@H](O)CC[C@]12C
InChI Key InChIKey=QGXBDMJGAMFCBF-HLUDHZFRSA-N
PDB links: 3 PDB IDs match this monomer.
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 17639
Found 3 hits for monomerid = 17639
Affinity DataIC50: 4.07E+4nMAssay Description:Inhibitory concentration against recombinant rat androgen receptor expressed in Escherichia coli using [3H]methyltrienolone (R 1881)More data for this Ligand-Target Pair
TargetGlucose-6-phosphate 1-dehydrogenase(Homo sapiens (Human))
University of Manchester
Curated by ChEMBL
University of Manchester
Curated by ChEMBL
Affinity DataIC50: 2.00E+5nMAssay Description:Inhibition of human recombinant N-terminal His tagged G6PD expressed in Escherichia coli JM109 (DE3) cells assessed as production of NADPH after 30 m...More data for this Ligand-Target Pair
Target17-beta-hydroxysteroid dehydrogenase type 3(Rattus norvegicus)
CHU de Qu£bec-Research Center (CHUL)
Curated by ChEMBL
CHU de Qu£bec-Research Center (CHUL)
Curated by ChEMBL
Affinity DataIC50: 90nMAssay Description:Inhibition of 17beta-HSD3 in rat testes microsomes using [14C]-4-androstene-3,17-dione as substrate after 2 hrsMore data for this Ligand-Target Pair