

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM111452 N-(4-bromophenyl)-4-methyl-3-(4-(pyridin-3-yl) pyrimidin-2-ylamino) benzamide (A5)
SMILES: Cc1ccc(cc1Nc1nccc(n1)-c1cccnc1)C(=O)Nc1ccc(Br)cc1
InChI Key: InChIKey=JQOGSOAHVQODHB-UHFFFAOYSA-N
Data: 1 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bcr-Abl (Homo sapiens (Human)) | BDBM111452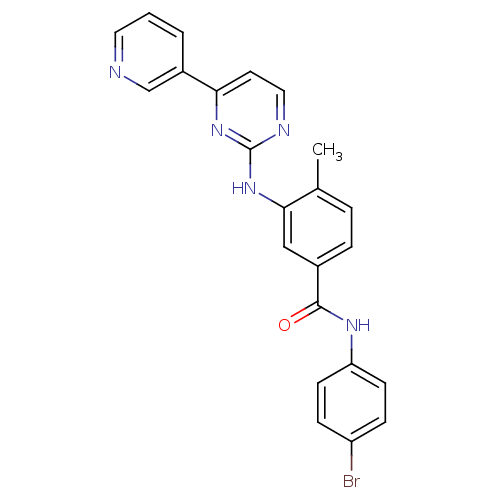 (N-(4-bromophenyl)-4-methyl-3-(4-(pyridin-3-yl) pyr...) | PDB MMDB Reactome pathway B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 483 | n/a | n/a | n/a | n/a | n/a | n/a |
Xi'an Jiaotong University, No. 76, Yanta West Road, Xi'an, 710061, China | Assay Description ATP (6.021 μM) and substrate (457.7 nM) were used. Kinase, substrate peptide, and inhibitor were added in a 384-well plate. The reaction was s... | Chem Biol Drug Des 83: 592-9 (2014) Article DOI: 10.1111/cbdd.12272 BindingDB Entry DOI: 10.7270/Q2GX497N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||