 Found 9 hits for monomerid = 14059
Found 9 hits for monomerid = 14059 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Homo sapiens (Human)) | BDBM14059
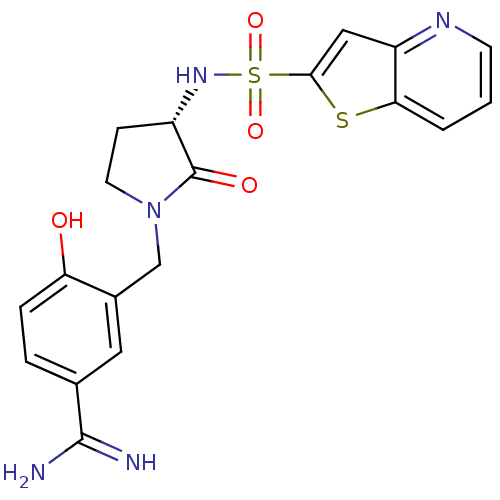
(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.700 | -12.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
J Med Chem 43: 3226-32 (2000)
Article DOI: 10.1021/jm000940u
BindingDB Entry DOI: 10.7270/Q25H7DHP |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Binding affinity against serine protease factor Xa (fXa) |
Bioorg Med Chem Lett 9: 2753-8 (1999)
BindingDB Entry DOI: 10.7270/Q2W66K02 |
More data for this
Ligand-Target Pair | |
Tissue-type plasminogen activator
(Homo sapiens (Human)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Binding affinity against tissue plasminogen activator (t-Pa) |
Bioorg Med Chem Lett 9: 2753-8 (1999)
BindingDB Entry DOI: 10.7270/Q2W66K02 |
More data for this
Ligand-Target Pair | |
Trypsin-1
(Homo sapiens (Human)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Binding affinity against trypsin |
Bioorg Med Chem Lett 9: 2753-8 (1999)
BindingDB Entry DOI: 10.7270/Q2W66K02 |
More data for this
Ligand-Target Pair | |
Trypsin
(Bos taurus (bovine)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 1.00E+3 | -8.10 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
J Med Chem 43: 3226-32 (2000)
Article DOI: 10.1021/jm000940u
BindingDB Entry DOI: 10.7270/Q25H7DHP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Plasminogen
(Homo sapiens (Human)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Binding affinity against plasmin |
Bioorg Med Chem Lett 9: 2753-8 (1999)
BindingDB Entry DOI: 10.7270/Q2W66K02 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| >4.00E+3 | >-7.28 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
J Med Chem 43: 3226-32 (2000)
Article DOI: 10.1021/jm000940u
BindingDB Entry DOI: 10.7270/Q25H7DHP |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Binding affinity against thrombin |
Bioorg Med Chem Lett 9: 2753-8 (1999)
BindingDB Entry DOI: 10.7270/Q2W66K02 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM14059

(4-hydroxy-3-[[(3S)-2-oxo-3-(9-thia-5-azabicyclo[4....)Show SMILES NC(=N)c1ccc(O)c(CN2CC[C@H](NS(=O)(=O)c3cc4ncccc4s3)C2=O)c1 |r| Show InChI InChI=1S/C19H19N5O4S2/c20-18(21)11-3-4-15(25)12(8-11)10-24-7-5-13(19(24)26)23-30(27,28)17-9-14-16(29-17)2-1-6-22-14/h1-4,6,8-9,13,23,25H,5,7,10H2,(H3,20,21)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 5.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Binding affinity against activated protein C (APC) |
Bioorg Med Chem Lett 9: 2753-8 (1999)
BindingDB Entry DOI: 10.7270/Q2W66K02 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data