

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM35730 cid_694792::sulfonamide deriv., 5e
SMILES: NS(=O)(=O)c1ccc(CNC(=O)Cc2cccs2)cc1
InChI Key: InChIKey=HFRDTVPDZVGHGW-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM35730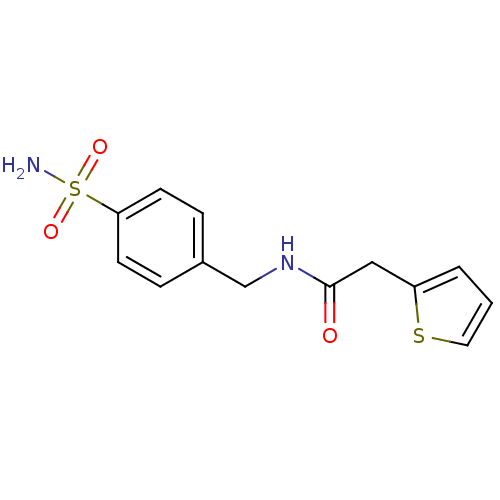 (cid_694792 | sulfonamide deriv., 5e) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 7.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istanbul University Curated by ChEMBL | Assay Description Inhibition of human recombinant carbonic anhydrase 7 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 19: 3170-3 (2009) Article DOI: 10.1016/j.bmcl.2009.04.123 BindingDB Entry DOI: 10.7270/Q2TQ61V6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VB (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 7.20 | -11.1 | n/a | n/a | n/a | n/a | n/a | n/a | 25 |
Istanbul University | Assay Description An SX.18MV-R Applied Photophysics (Oxford, UK) stopped-flow instrument has been used to assay the catalytic/inhibition of various CA isozymes. Phenol... | Bioorg Med Chem 17: 4894-9 (2009) Article DOI: 10.1016/j.bmc.2009.06.006 BindingDB Entry DOI: 10.7270/Q2RJ4GT1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VA (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 9.10 | -11.0 | n/a | n/a | n/a | n/a | n/a | n/a | 25 |
Istanbul University | Assay Description An SX.18MV-R Applied Photophysics (Oxford, UK) stopped-flow instrument has been used to assay the catalytic/inhibition of various CA isozymes. Phenol... | Bioorg Med Chem 17: 4894-9 (2009) Article DOI: 10.1016/j.bmc.2009.06.006 BindingDB Entry DOI: 10.7270/Q2RJ4GT1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istanbul University Curated by ChEMBL | Assay Description Inhibition of human recombinant carbonic anhydrase 2 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 19: 3170-3 (2009) Article DOI: 10.1016/j.bmcl.2009.04.123 BindingDB Entry DOI: 10.7270/Q2TQ61V6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 52 | -9.93 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Istanbul University | Assay Description An SX.18MV-R Applied Photophysics (Oxford, UK) stopped-flow instrument has been used to assay the catalytic/inhibition of various CA isozymes. Phenol... | Bioorg Med Chem 17: 4894-9 (2009) Article DOI: 10.1016/j.bmc.2009.06.006 BindingDB Entry DOI: 10.7270/Q2RJ4GT1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 60 | -9.85 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Istanbul University | Assay Description An SX.18MV-R Applied Photophysics (Oxford, UK) stopped-flow instrument has been used to assay the catalytic/inhibition of various CA isozymes. Phenol... | Bioorg Med Chem 17: 4894-9 (2009) Article DOI: 10.1016/j.bmc.2009.06.006 BindingDB Entry DOI: 10.7270/Q2RJ4GT1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Istanbul University Curated by ChEMBL | Assay Description Inhibition of human recombinant carbonic anhydrase 1 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 19: 3170-3 (2009) Article DOI: 10.1016/j.bmcl.2009.04.123 BindingDB Entry DOI: 10.7270/Q2TQ61V6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Hematopoietic cell protein-tyrosine phosphatase 70Z-PEP (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | PCBioAssay | n/a | n/a | 5.61E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Center for Chemical Genomics Curated by PubChem BioAssay | PubChem Bioassay (2013) BindingDB Entry DOI: 10.7270/Q2DN43N5 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Low molecular weight phosphotyrosine protein phosphatase (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | PCBioAssay | n/a | n/a | >8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Center for Chemical Genomics Curated by PubChem BioAssay | PubChem Bioassay (2013) BindingDB Entry DOI: 10.7270/Q2JD4VDZ | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Low molecular weight phosphotyrosine protein phosphatase (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | PCBioAssay | n/a | n/a | 2.07E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Center for Chemical Genomics Curated by PubChem BioAssay | PubChem Bioassay (2012) BindingDB Entry DOI: 10.7270/Q2GM85XC | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Voltage-gated calcium channel subunit alpha Cav2.2 (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | PCBioAssay | n/a | n/a | 3.86E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Center for Chemical Genomics Curated by PubChem BioAssay | Assay Description Data Source: Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute Network... | PubChem Bioassay (2009) BindingDB Entry DOI: 10.7270/Q2PZ578V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| core protein (Hepatitis C virus) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | PCBioAssay | n/a | n/a | 4.92E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute Molecular Screening Center Curated by PubChem BioAssay | Assay Description Source (MLPCN Center Name): The Scripps Research Institute Molecular Screening Center Affiliation: The Scripps Research Institute, TSRI Assay Provide... | PubChem Bioassay (2009) BindingDB Entry DOI: 10.7270/Q29Z939R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| carboxy-terminal domain RNA polymerase II polypeptide A small phosphatase 1 isoform 1 (Homo sapiens (Human)) | BDBM35730 (cid_694792 | sulfonamide deriv., 5e) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | PCBioAssay | n/a | n/a | 4.18E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Burnham Center for Chemical Genomics Curated by PubChem BioAssay | Assay Description Data Source: Sanford-Burnham Center for Chemical Genomics (SBCCG) Source Affiliation: Sanford-Burnham Medical Research Institute (SBMRI, San Diego, C... | PubChem Bioassay (2011) BindingDB Entry DOI: 10.7270/Q2V986KM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||