

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50062439 9-Chloro-5-(4-chloro-3-methyl-phenyl)-2,2,4-trimethyl-2,5-dihydro-1H-6-oxa-1-aza-chrysene::CHEMBL94753
SMILES: CC1=CC(C)(C)Nc2ccc3-c4cc(Cl)ccc4OC(c4ccc(Cl)c(C)c4)c3c12
InChI Key: InChIKey=SSFWXDSRZJERKU-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Progesterone receptor (Homo sapiens (Human)) | BDBM50062439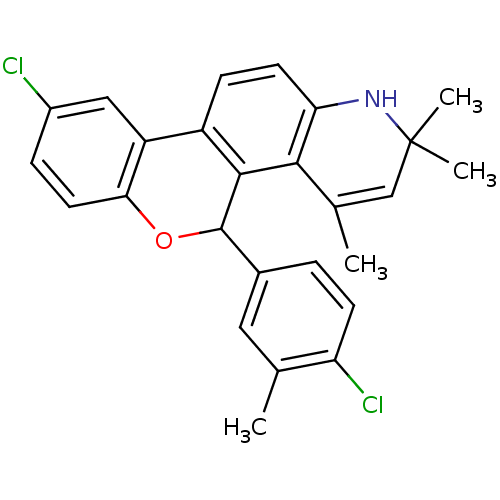 (9-Chloro-5-(4-chloro-3-methyl-phenyl)-2,2,4-trimet...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description The binding affinity measured using baculovirus-expressed hPR-A in sf21 cells. | J Med Chem 41: 303-10 (1998) Article DOI: 10.1021/jm9705770 BindingDB Entry DOI: 10.7270/Q2RV0PC8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Progesterone receptor (Homo sapiens (Human)) | BDBM50062439 (9-Chloro-5-(4-chloro-3-methyl-phenyl)-2,2,4-trimet...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals Inc. Curated by ChEMBL | Assay Description Binding affinity for human progesterone receptor isoform A expressed in CV-1 cells | J Med Chem 41: 2779-85 (1998) Article DOI: 10.1021/jm980190c BindingDB Entry DOI: 10.7270/Q2CV4JFM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glucocorticoid receptor (Homo sapiens (Human)) | BDBM50062439 (9-Chloro-5-(4-chloro-3-methyl-phenyl)-2,2,4-trimet...) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals Inc. Curated by ChEMBL | Assay Description Binding affinity for human glucocorticoid receptor expressed in CV-1 cells | J Med Chem 41: 2779-85 (1998) Article DOI: 10.1021/jm980190c BindingDB Entry DOI: 10.7270/Q2CV4JFM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Progesterone receptor (Homo sapiens (Human)) | BDBM50062439 (9-Chloro-5-(4-chloro-3-methyl-phenyl)-2,2,4-trimet...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 9.40 | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals Inc. Curated by ChEMBL | Assay Description Effective concentration for half-maximal activation of human progesterone receptor expressed in CV-1 cells | J Med Chem 41: 2779-85 (1998) Article DOI: 10.1021/jm980190c BindingDB Entry DOI: 10.7270/Q2CV4JFM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Progesterone receptor (Homo sapiens (Human)) | BDBM50062439 (9-Chloro-5-(4-chloro-3-methyl-phenyl)-2,2,4-trimet...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 9.40 | n/a | n/a | n/a | n/a |
Ligand Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Agonistic activity was measured for modulation of hPR-B (human progesterone receptor) in co-transfected CV-1 cells. | J Med Chem 41: 303-10 (1998) Article DOI: 10.1021/jm9705770 BindingDB Entry DOI: 10.7270/Q2RV0PC8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||