 Found 7 hits for monomerid = 50076995
Found 7 hits for monomerid = 50076995 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50076995
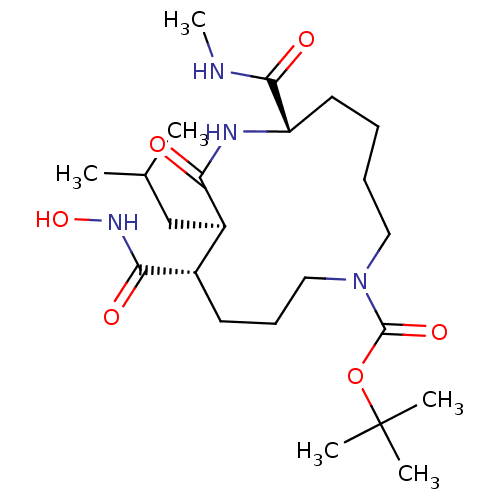
((6S,9R,10S)-10-Hydroxycarbamoyl-9-isobutyl-6-methy...)Show SMILES CNC(=O)[C@@H]1CCCCN(CCC[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)C(=O)OC(C)(C)C Show InChI InChI=1S/C23H42N4O6/c1-15(2)14-17-16(20(29)26-32)10-9-13-27(22(31)33-23(3,4)5)12-8-7-11-18(21(30)24-6)25-19(17)28/h15-18,32H,7-14H2,1-6H3,(H,24,30)(H,25,28)(H,26,29)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceutical Co.
Curated by ChEMBL
| Assay Description
Inhibition of MMP-9 (matrix metalloprotease-9, gelatinase B) |
Bioorg Med Chem Lett 9: 1279-84 (1999)
BindingDB Entry DOI: 10.7270/Q2T43TK4 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50076995

((6S,9R,10S)-10-Hydroxycarbamoyl-9-isobutyl-6-methy...)Show SMILES CNC(=O)[C@@H]1CCCCN(CCC[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)C(=O)OC(C)(C)C Show InChI InChI=1S/C23H42N4O6/c1-15(2)14-17-16(20(29)26-32)10-9-13-27(22(31)33-23(3,4)5)12-8-7-11-18(21(30)24-6)25-19(17)28/h15-18,32H,7-14H2,1-6H3,(H,24,30)(H,25,28)(H,26,29)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of human MMP-9. |
J Med Chem 44: 2636-60 (2001)
BindingDB Entry DOI: 10.7270/Q2FB53NR |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50076995

((6S,9R,10S)-10-Hydroxycarbamoyl-9-isobutyl-6-methy...)Show SMILES CNC(=O)[C@@H]1CCCCN(CCC[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)C(=O)OC(C)(C)C Show InChI InChI=1S/C23H42N4O6/c1-15(2)14-17-16(20(29)26-32)10-9-13-27(22(31)33-23(3,4)5)12-8-7-11-18(21(30)24-6)25-19(17)28/h15-18,32H,7-14H2,1-6H3,(H,24,30)(H,25,28)(H,26,29)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of human MMP-1. |
J Med Chem 44: 2636-60 (2001)
BindingDB Entry DOI: 10.7270/Q2FB53NR |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50076995

((6S,9R,10S)-10-Hydroxycarbamoyl-9-isobutyl-6-methy...)Show SMILES CNC(=O)[C@@H]1CCCCN(CCC[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)C(=O)OC(C)(C)C Show InChI InChI=1S/C23H42N4O6/c1-15(2)14-17-16(20(29)26-32)10-9-13-27(22(31)33-23(3,4)5)12-8-7-11-18(21(30)24-6)25-19(17)28/h15-18,32H,7-14H2,1-6H3,(H,24,30)(H,25,28)(H,26,29)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceutical Co.
Curated by ChEMBL
| Assay Description
Inhibition of MMP-1 (human fibroblast collagenase) |
Bioorg Med Chem Lett 9: 1279-84 (1999)
BindingDB Entry DOI: 10.7270/Q2T43TK4 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50076995

((6S,9R,10S)-10-Hydroxycarbamoyl-9-isobutyl-6-methy...)Show SMILES CNC(=O)[C@@H]1CCCCN(CCC[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)C(=O)OC(C)(C)C Show InChI InChI=1S/C23H42N4O6/c1-15(2)14-17-16(20(29)26-32)10-9-13-27(22(31)33-23(3,4)5)12-8-7-11-18(21(30)24-6)25-19(17)28/h15-18,32H,7-14H2,1-6H3,(H,24,30)(H,25,28)(H,26,29)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceutical Co.
Curated by ChEMBL
| Assay Description
Inhibition of MMP-3 (human fibroblast stromelysin) |
Bioorg Med Chem Lett 9: 1279-84 (1999)
BindingDB Entry DOI: 10.7270/Q2T43TK4 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50076995

((6S,9R,10S)-10-Hydroxycarbamoyl-9-isobutyl-6-methy...)Show SMILES CNC(=O)[C@@H]1CCCCN(CCC[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)C(=O)OC(C)(C)C Show InChI InChI=1S/C23H42N4O6/c1-15(2)14-17-16(20(29)26-32)10-9-13-27(22(31)33-23(3,4)5)12-8-7-11-18(21(30)24-6)25-19(17)28/h15-18,32H,7-14H2,1-6H3,(H,24,30)(H,25,28)(H,26,29)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Inhibition of human MMP-3. |
J Med Chem 44: 2636-60 (2001)
BindingDB Entry DOI: 10.7270/Q2FB53NR |
More data for this
Ligand-Target Pair | |
Disintegrin and metalloproteinase domain-containing protein 17
(Homo sapiens (Human)) | BDBM50076995

((6S,9R,10S)-10-Hydroxycarbamoyl-9-isobutyl-6-methy...)Show SMILES CNC(=O)[C@@H]1CCCCN(CCC[C@@H]([C@@H](CC(C)C)C(=O)N1)C(=O)NO)C(=O)OC(C)(C)C Show InChI InChI=1S/C23H42N4O6/c1-15(2)14-17-16(20(29)26-32)10-9-13-27(22(31)33-23(3,4)5)12-8-7-11-18(21(30)24-6)25-19(17)28/h15-18,32H,7-14H2,1-6H3,(H,24,30)(H,25,28)(H,26,29)/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
DuPont Pharmaceuticals Company
Curated by ChEMBL
| Assay Description
Inhibition of LPS-stimulated TNF-alpha release in human whole blood |
J Med Chem 44: 2636-60 (2001)
BindingDB Entry DOI: 10.7270/Q2FB53NR |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data