

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50079824 ((S)-1-{(S)-1-[(S)-(S)-1-(1-Carbamoyl-3-methyl-butylcarbamoyl)-butylcarbamoyl]-4-guanidino-butylcarbamoyl}-2-phenyl-ethyl)-carbamic acid ethyl ester::((S)-1-{(S)-1-[(S)-1-((S)-1-Carbamoyl-3-methyl-butylcarbamoyl)-butylcarbamoyl]-4-guanidino-butylcarbamoyl}-2-phenyl-ethyl)-carbamic acid ethyl ester::CHEMBL292340
SMILES: CCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1ccccc1)NC(=O)OCC)C(=O)N[C@@H](CC(C)C)C(N)=O
InChI Key: InChIKey=BYJYKJYYOSDFFJ-MLCQCVOFSA-N
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HLA class II histocompatibility antigen DRB1-1 (Homo sapiens (Human)) | BDBM50079824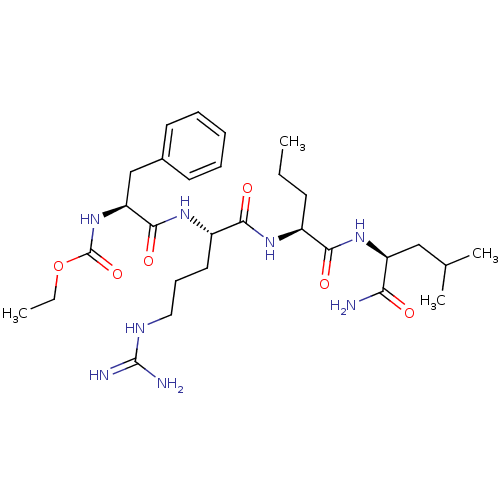 (((S)-1-{(S)-1-[(S)-(S)-1-(1-Carbamoyl-3-methyl-but...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory concentration to inhibit the binding of biotinylated rat myelin basic protein peptide (RMBP90-102) against DR1 allele of class II MHC for ... | Bioorg Med Chem Lett 7: 19-24 (1997) Article DOI: 10.1016/S0960-894X(96)00579-3 BindingDB Entry DOI: 10.7270/Q2Q52PMZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human leukocyte antigen DR beta chain (Homo sapiens (Human)) | BDBM50079824 (((S)-1-{(S)-1-[(S)-(S)-1-(1-Carbamoyl-3-methyl-but...) | PDB MMDB NCI pathway Reactome pathway UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory concentration required to inhibit the binding of biotinylated rat myelin basic protein peptide (RMBP90-102) against DR1 allele of class II... | Bioorg Med Chem Lett 7: 19-24 (1997) Article DOI: 10.1016/S0960-894X(96)00579-3 BindingDB Entry DOI: 10.7270/Q2Q52PMZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| MHC class II (Rattus norvegicus) | BDBM50079824 (((S)-1-{(S)-1-[(S)-(S)-1-(1-Carbamoyl-3-methyl-but...) | PDB Reactome pathway KEGG UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Ability to inhibit binding of biotinylated rat myelin basic protein 13-mer peptide (RMBP 90-102, Major histocompatibility complex class II to purifie... | Bioorg Med Chem Lett 9: 2109-14 (1999) BindingDB Entry DOI: 10.7270/Q2NZ86TF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||