

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: [NH3+][C@H](c1ccccc1)P(O)(=O)C[C@@H](Cc1ccc(cc1)-c1ccccc1)C(=O)NCC(O)=O
InChI Key: InChIKey=NIIDEJBIXSLRGB-VWNXMTODSA-O
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neprilysin (Oryctolagus cuniculus (rabbit)) | BDBM50087104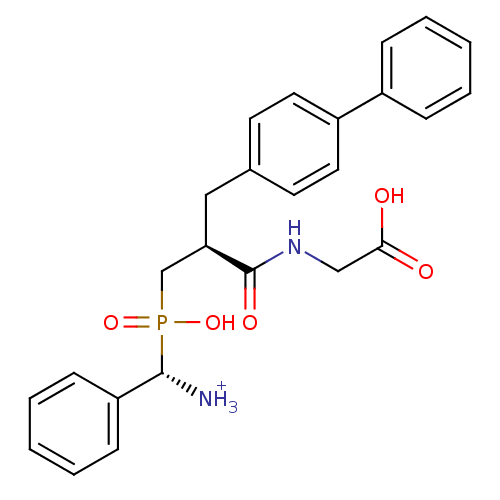 (C-{[3-Biphenyl-4-yl-2-(carboxymethyl-carbamoyl)-pr...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on neutral endopeptidase (NEP) using DGNPA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aminopeptidase N (Sus scrofa (Pig)) | BDBM50087104 (C-{[3-Biphenyl-4-yl-2-(carboxymethyl-carbamoyl)-pr...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 32.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description Inhibitory activity on aminopeptidase N (APN) using Ala-pNA as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Angiotensin-converting enzyme (Rattus norvegicus) | BDBM50087104 (C-{[3-Biphenyl-4-yl-2-(carboxymethyl-carbamoyl)-pr...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | PubMed | 7.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM Curated by ChEMBL | Assay Description inhibitory activity on angiotensin I converting enzyme (ACE) using cbz-Phe-His-Leu as substrate. | J Med Chem 43: 1398-408 (2001) BindingDB Entry DOI: 10.7270/Q2TQ627J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||