

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50199176 2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-dione 5'-triphosphate::CHEMBL217803
SMILES: O[C@H]1[C@@H](O)[C@@H](O[C@@H]1COP(O)(=O)OP(O)(=O)OP(O)(O)=O)n1ccc(=O)[nH]c1=S
InChI Key: InChIKey=KHYOUGAATNYCAZ-XVFCMESISA-N
Data: 12 EC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Purinergic receptor P2Y2 (Homo sapiens (Human)) | BDBM50199176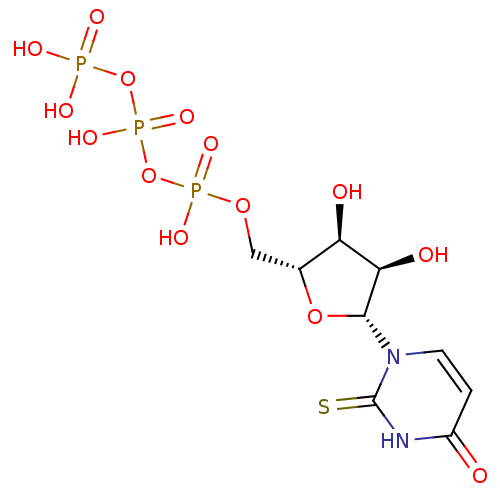 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Agonist activity at human recombinant P2Y2 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate production after 30 mins by sc... | J Med Chem 54: 4018-33 (2011) Article DOI: 10.1021/jm101591j BindingDB Entry DOI: 10.7270/Q22F7NT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y4 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 350 | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Agonist activity at human P2Y4 | J Med Chem 54: 4018-33 (2011) Article DOI: 10.1021/jm101591j BindingDB Entry DOI: 10.7270/Q22F7NT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y6 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Agonist activity at human recombinant P2Y6 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate production after 30 mins by sc... | J Med Chem 54: 4018-33 (2011) Article DOI: 10.1021/jm101591j BindingDB Entry DOI: 10.7270/Q22F7NT0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y6 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Agonist activity at human P2Y6 receptor expressed in 1321N1 cells assessed as IP accumulation by SPA | J Med Chem 49: 7076-87 (2006) Article DOI: 10.1021/jm060848j BindingDB Entry DOI: 10.7270/Q2SN09RW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Purinergic receptor P2Y2 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 50 | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Agonist activity at human P2Y2 receptor expressed in 1321N1 cells assessed as IP accumulation by SPA | J Med Chem 49: 7076-87 (2006) Article DOI: 10.1021/jm060848j BindingDB Entry DOI: 10.7270/Q2SN09RW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y6 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Agonist activity at human P2Y6 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate accumulation by scintillation proximity as... | Bioorg Med Chem 16: 6319-32 (2008) Article DOI: 10.1016/j.bmc.2008.05.013 BindingDB Entry DOI: 10.7270/Q2K07569 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Purinergic receptor P2Y2 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Agonist activity at human recombinant P2Y2 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase C | J Med Chem 50: 1166-76 (2007) Article DOI: 10.1021/jm060903o BindingDB Entry DOI: 10.7270/Q2PK0GZ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y4 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 350 | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Agonist activity at human recombinant P2Y4 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase C | J Med Chem 50: 1166-76 (2007) Article DOI: 10.1021/jm060903o BindingDB Entry DOI: 10.7270/Q2PK0GZ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y6 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases Curated by ChEMBL | Assay Description Agonist activity at human recombinant P2Y6 receptor expressed in 1321N1 cells assessed as stimulation of phospholipase C | J Med Chem 50: 1166-76 (2007) Article DOI: 10.1021/jm060903o BindingDB Entry DOI: 10.7270/Q2PK0GZ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Purinergic receptor P2Y2 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Agonist activity at human P2Y2 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate accumulation by scintillation proximity as... | Bioorg Med Chem 16: 6319-32 (2008) Article DOI: 10.1016/j.bmc.2008.05.013 BindingDB Entry DOI: 10.7270/Q2K07569 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y4 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 350 | n/a | n/a | n/a | n/a |
NIDDK Curated by ChEMBL | Assay Description Agonist activity at human P2Y4 receptor expressed in human 1321N1 cells assessed as [3H]inositol phosphate accumulation by scintillation proximity as... | Bioorg Med Chem 16: 6319-32 (2008) Article DOI: 10.1016/j.bmc.2008.05.013 BindingDB Entry DOI: 10.7270/Q2K07569 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pyrimidinergic receptor P2Y4 (Homo sapiens (Human)) | BDBM50199176 (2-thio-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-di...) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.77E+3 | n/a | n/a | n/a | n/a |
University of Bonn Curated by ChEMBL | Assay Description Agonist activity at human P2Y4 receptor expressed in 1321N1 cells assessed as IP accumulation by SPA | J Med Chem 49: 7076-87 (2006) Article DOI: 10.1021/jm060848j BindingDB Entry DOI: 10.7270/Q2SN09RW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||