

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50209568 4',7,8-trihydroxyisoflavone::7,8,4'-trihydroxyisoflavone::7,8-dihydroxy-3-(4-hydroxyphenyl)-4H-chromen-4-one::CHEMBL242739::IR-301
SMILES: Oc1ccc(cc1)-c1coc2c(O)c(O)ccc2c1=O
InChI Key: InChIKey=BMZFZTMWBCFKSS-UHFFFAOYSA-N
Data: 10 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Arachidonate 5-lipoxygenase (Homo sapiens (Human)) | BDBM50209568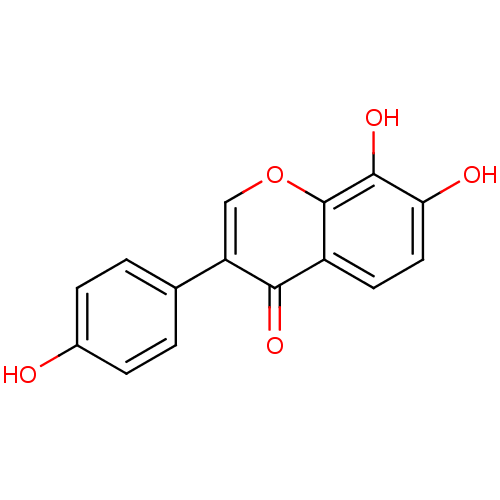 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 900 | n/a | n/a | n/a | n/a | 7.3 | 25 |
Universidad de Santiago | Assay Description The reactions were done in a volume of 2 mL and constantly stirred using a magnetic stir bar at room temperature (23 �C). Reactions with the crude, a... | Chem Biol Drug Des 86: 114-21 (2015) Article DOI: 10.1111/cbdd.12469 BindingDB Entry DOI: 10.7270/Q2PZ57JF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin G/H synthase (cyclooxygenase) (Ovis aries (Sheep)) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.50E+5 | n/a | n/a | n/a | n/a | 8.0 | n/a |
Universidad de Santiago | Assay Description Approximately 2 �g of either COX-1 or COX-2 was added to buffer containing 100 �M AA, 0.1 M Tris-HCl buffer (pH 8.0), 5 mM EDTA, 2 mM phenol, and 1 �... | Chem Biol Drug Des 86: 114-21 (2015) Article DOI: 10.1111/cbdd.12469 BindingDB Entry DOI: 10.7270/Q2PZ57JF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclooxygenase (Homo sapiens (Human)) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.50E+5 | n/a | n/a | n/a | n/a | 8.0 | n/a |
Universidad de Santiago | Assay Description Approximately 2 �g of either COX-1 or COX-2 was added to buffer containing 100 �M AA, 0.1 M Tris-HCl buffer (pH 8.0), 5 mM EDTA, 2 mM phenol, and 1 �... | Chem Biol Drug Des 86: 114-21 (2015) Article DOI: 10.1111/cbdd.12469 BindingDB Entry DOI: 10.7270/Q2PZ57JF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Xanthine dehydrogenase/oxidase (Homo sapiens (Human)) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 6.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AmorePacific Corporation Curated by ChEMBL | Assay Description Inhibition of xanthine oxidase (unknown origin) assessed as decrease of superoxide generation | Bioorg Med Chem Lett 18: 5006-9 (2008) Article DOI: 10.1016/j.bmcl.2008.08.016 BindingDB Entry DOI: 10.7270/Q2QF8SQK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urease subunit beta (Helicobacter pylori (strain ATCC 700392 / 26695) (...) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.48E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University Curated by ChEMBL | Assay Description Inhibition of Helicobacter pylori ATCC 43504 urease in presence of 0.4 mM 2-mercaptoethanol after 3 hrs pre-incubation | Bioorg Med Chem 15: 3703-10 (2007) Article DOI: 10.1016/j.bmc.2007.03.045 BindingDB Entry DOI: 10.7270/Q2P55R84 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosinase (Mus musculus (Mouse)) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | UniProtKB/SwissProt UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AmorePacific Corporation Curated by ChEMBL | Assay Description Inhibition of tyrosinase in mouse Melan-a cells by ELISA | Bioorg Med Chem Lett 20: 1162-4 (2010) Article DOI: 10.1016/j.bmcl.2009.12.021 BindingDB Entry DOI: 10.7270/Q2M045JZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 3 (PFKFB3) (Homo sapiens (Human)) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 670 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Auckland Curated by ChEMBL | Assay Description Inhibition of PFKFB3 (unknown origin) | Bioorg Med Chem 22: 1029-39 (2014) Article DOI: 10.1016/j.bmc.2013.12.041 BindingDB Entry DOI: 10.7270/Q2TB18C4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urease subunit beta (Helicobacter pylori (strain ATCC 700392 / 26695) (...) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University Curated by ChEMBL | Assay Description Inhibition of Helicobacter pylori ATCC 43504 urease after 3 hrs pre-incubation | Bioorg Med Chem 15: 3703-10 (2007) Article DOI: 10.1016/j.bmc.2007.03.045 BindingDB Entry DOI: 10.7270/Q2P55R84 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urease subunit beta (Helicobacter pylori (strain ATCC 700392 / 26695) (...) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.48E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University Curated by ChEMBL | Assay Description Inhibition of Helicobacter pylori ATCC 43504 urease in presence of 0.4 mM dithiothreitol after 3 hrs pre-incubation | Bioorg Med Chem 15: 3703-10 (2007) Article DOI: 10.1016/j.bmc.2007.03.045 BindingDB Entry DOI: 10.7270/Q2P55R84 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Xanthine dehydrogenase/oxidase (Homo sapiens (Human)) | BDBM50209568 (4',7,8-trihydroxyisoflavone | 7,8,4'-trihydroxyiso...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
AmorePacific Corporation Curated by ChEMBL | Assay Description Inhibition of xanthine oxidase (unknown origin) assessed as decrease of uric acid generation | Bioorg Med Chem Lett 18: 5006-9 (2008) Article DOI: 10.1016/j.bmcl.2008.08.016 BindingDB Entry DOI: 10.7270/Q2QF8SQK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||