

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50269614 2-Methyl-beta-carboline-2-ium iodide::2-methyl-2H-pyrido[3,4-b]indole hydroiodide::2-methylnorharmane iodide::CHEMBL455600
SMILES: C[n+]1ccc2c(c1)[nH]c1ccccc21
InChI Key: InChIKey=UDHBHTHWWOQPBW-UHFFFAOYSA-O
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Butyrylcholinesterase (BuChE) (Equus caballus (Horse)) | BDBM50269614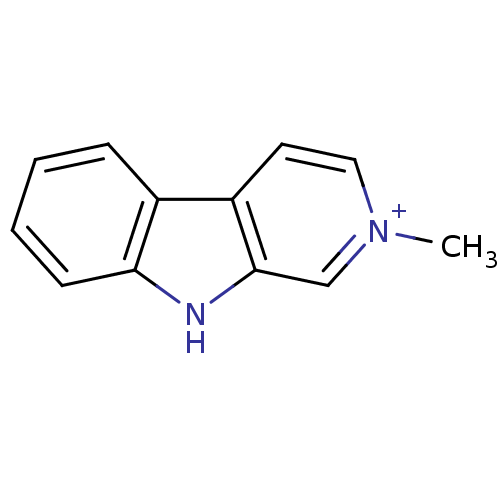 (2-Methyl-beta-carboline-2-ium iodide | 2-methyl-2H...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Zürich Curated by ChEMBL | Assay Description Inhibition of horse serum BChE by Ellman's reaction | J Nat Prod 68: 1793-5 (2005) Article DOI: 10.1021/np050312l BindingDB Entry DOI: 10.7270/Q2K9378M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50269614 (2-Methyl-beta-carboline-2-ium iodide | 2-methyl-2H...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Friedrich-Schiller-Universitat Jena Curated by ChEMBL | Assay Description Inhibition of electric eel AChE by modified Ellman's method | J Med Chem 53: 3611-7 (2010) Article DOI: 10.1021/jm1000024 BindingDB Entry DOI: 10.7270/Q2SJ1KS0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Butyrylcholinesterase (BuChE) (Equus caballus (Horse)) | BDBM50269614 (2-Methyl-beta-carboline-2-ium iodide | 2-methyl-2H...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 540 | n/a | n/a | n/a | n/a | n/a | n/a |
Friedrich-Schiller-Universitat Jena Curated by ChEMBL | Assay Description Inhibition of equine serum BChE by modified Ellman's method | J Med Chem 53: 3611-7 (2010) Article DOI: 10.1021/jm1000024 BindingDB Entry DOI: 10.7270/Q2SJ1KS0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||