

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50330016 ((3R,5R)-4-(4-chlorophenylsulfonyl)-5-(1-methylcyclopropyl)morpholin-3-yl)methyl 4-(1-hydroxy-2-methylpropan-2-yl)piperazine-1-carboxylate::CHEMBL1271822
SMILES: CC(C)(CO)N1CCN(CC1)C(=O)OC[C@H]1COC[C@H](N1S(=O)(=O)c1ccc(Cl)cc1)C1(C)CC1
InChI Key: InChIKey=MBQIBPDSTIZHQM-CTNGQTDRSA-N
Data: 1 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cytochrome P450 3A4 (Homo sapiens (Human)) | BDBM50330016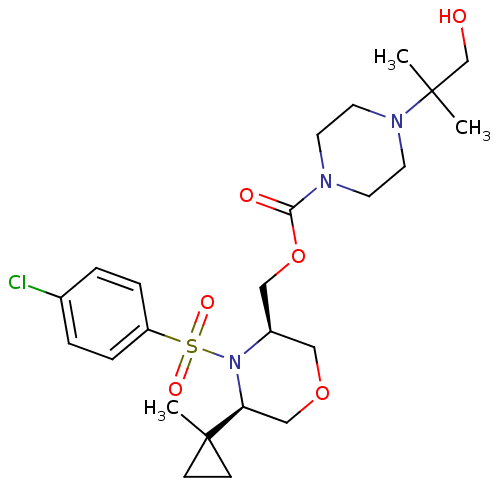 (((3R,5R)-4-(4-chlorophenylsulfonyl)-5-(1-methylcyc...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Lab. Curated by ChEMBL | Assay Description Inhibition of CYP3A4 after 30 mins | Bioorg Med Chem Lett 20: 6606-9 (2010) Article DOI: 10.1016/j.bmcl.2010.09.028 BindingDB Entry DOI: 10.7270/Q2RF5V79 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||