

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50352643 CHEMBL1822699
SMILES: CCN(CC)c1ccc2c(Oc3cc(ccc3C22N(C(=O)c3ccccc23)c2ccc(cc2)S(N)(=O)=O)N(CC)CC)c1
InChI Key: InChIKey=UDDMXLGFVDCGOV-UHFFFAOYSA-N
Data: 13 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic Anhydrase XIV (Homo sapiens (Human)) | BDBM50352643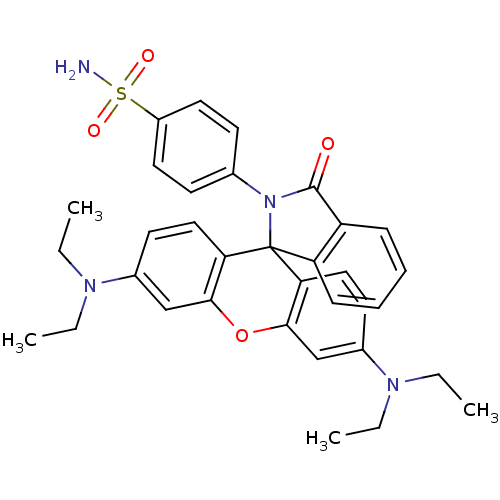 (CHEMBL1822699) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.70 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 14 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 9.10 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 12 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 18.5 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 9 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 60.2 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 7 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 279 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 2 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 13 (CA XIII) (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 280 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 13 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VB (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 310 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 5B preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase VA (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 413 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 5A preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 512 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 1 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 6 (CA-VI) (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 980 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 6 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| beta-Carbonic Anhydrase (Candida albicans (Yeast)) | BDBM50352643 (CHEMBL1822699) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.99E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 8.3 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of Candida albicans carbonic anhydrase preincubated for 15 mins at pH 8.3 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 4 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic Anhydrase III (Homo sapiens (Human)) | BDBM50352643 (CHEMBL1822699) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Ecole Nationale Sup£rieure de Chimie de Montpellier Curated by ChEMBL | Assay Description Inhibition of human carbonic anhydrase 3 preincubated for 15 mins at pH 7.5 by stopped flow CO2 hydration method | Bioorg Med Chem Lett 21: 5210-3 (2011) Article DOI: 10.1016/j.bmcl.2011.07.045 BindingDB Entry DOI: 10.7270/Q2XP75B3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||