 Found 12 hits for monomerid = 50362839
Found 12 hits for monomerid = 50362839 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Aldo-keto reductase family member 1B10 (AKR1B10)
(Homo sapiens (Human)) | BDBM50362839
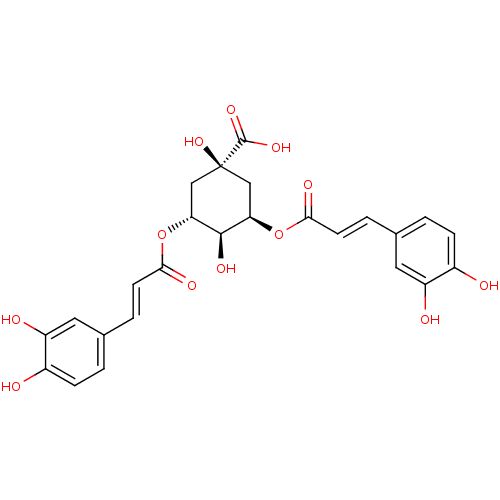
(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminus His6-tagged AKR1B10 expressed in Escherichia coli BL21 DE3 assessed as pyridine-3-aldehyde reduction by sp... |
Eur J Med Chem 48: 321-9 (2012)
Article DOI: 10.1016/j.ejmech.2011.12.034
BindingDB Entry DOI: 10.7270/Q2TT4RDB |
More data for this
Ligand-Target Pair | |
Histone deacetylase
(Homo sapiens (Human)) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 1.43E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Sungkyunkwan University
Curated by ChEMBL
| Assay Description
Inhibition of HDAC in human HeLa nuclear extract using fluor de lys as substrate after 10 to 15 mins by spectrofluorometry |
Bioorg Med Chem Lett 26: 2365-9 (2016)
BindingDB Entry DOI: 10.7270/Q2KH0Q6F |
More data for this
Ligand-Target Pair | |
Histone deacetylase
(Homo sapiens (Human)) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Sungkyunkwan University
Curated by ChEMBL
| Assay Description
Inhibition of HDAC in human HeLa cells using Boc-Lys(AC)-AMC as substrate after 24 to 48 hrs by spectrofluorometry |
Bioorg Med Chem Lett 26: 2365-9 (2016)
BindingDB Entry DOI: 10.7270/Q2KH0Q6F |
More data for this
Ligand-Target Pair | |
Aldose reductase (AR)
(Homo sapiens (Human)) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 88 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant N-terminus His6-tagged AKR1B1 expressed in Escherichia coli BL21 DE3 assessed as pyridine-3-aldehyde reduction by spe... |
Eur J Med Chem 48: 321-9 (2012)
Article DOI: 10.1016/j.ejmech.2011.12.034
BindingDB Entry DOI: 10.7270/Q2TT4RDB |
More data for this
Ligand-Target Pair | |
Human immunodeficiency virus type 1 integrase
(Human immunodeficiency virus 1) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Inhibitory activity against Human Immunodeficiency Virus Type 1 integrase (HIV-1 IN) in the disintegration assay. |
J Med Chem 42: 497-509 (1999)
Article DOI: 10.1021/jm9804735
BindingDB Entry DOI: 10.7270/Q2G161J5 |
More data for this
Ligand-Target Pair | |
Beta amyloid A4 protein
(Homo sapiens (Human)) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.64E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Inhibition of human amyloid beta (1 to 42) aggregation after 24 hrs by thioflavin-T fluorescence assay |
Bioorg Med Chem 20: 5844-9 (2012)
Article DOI: 10.1016/j.bmc.2012.08.001
BindingDB Entry DOI: 10.7270/Q2M048BH |
More data for this
Ligand-Target Pair | |
EBifunctional epoxide hydrolase 2
(Homo sapiens (Human)) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Chungnam National University
Curated by ChEMBL
| Assay Description
Inhibition of sEH (unknown origin) using PHOME as substrate after 40 mins by fluorescence photometric method |
Bioorg Med Chem Lett 27: 1874-1879 (2017)
Article DOI: 10.1016/j.bmcl.2017.02.013
BindingDB Entry DOI: 10.7270/Q21838SF |
More data for this
Ligand-Target Pair | |
Protein-tyrosine phosphatase 1B
(Homo sapiens (Human)) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.79E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B (unknown origin) |
Bioorg Med Chem Lett 28: 1194-1197 (2018)
Article DOI: 10.1016/j.bmcl.2018.02.052
BindingDB Entry DOI: 10.7270/Q20Z75V6 |
More data for this
Ligand-Target Pair | |
Integrase
(Human immunodeficiency virus 1) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.14E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science and Technology
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 recombinant integrase expressed in Escherichia coli |
J Nat Prod 69: 600-3 (2006)
Article DOI: 10.1021/np050447r
BindingDB Entry DOI: 10.7270/Q24F1THC |
More data for this
Ligand-Target Pair | |
Integrase
(Human immunodeficiency virus 1) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 integrase-catalyzed disintegration activity |
J Nat Prod 64: 265-77 (2001)
Article DOI: 10.1021/np0003995
BindingDB Entry DOI: 10.7270/Q2HT2S52 |
More data for this
Ligand-Target Pair | |
Integrase
(Human immunodeficiency virus 1) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 640 | n/a | n/a | n/a | n/a | n/a | n/a |
UMR CNRS 8009
Curated by ChEMBL
| Assay Description
Inhibition of HIV1 integrase catalyzed reactions |
Bioorg Med Chem Lett 18: 4736-40 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.063
BindingDB Entry DOI: 10.7270/Q2Q24311 |
More data for this
Ligand-Target Pair | |
Aldose reductase
(Rattus norvegicus) | BDBM50362839

(CHEMBL249447)Show SMILES O[C@H]1[C@@H](C[C@@](O)(C[C@H]1OC(=O)\C=C\c1ccc(O)c(O)c1)C(O)=O)OC(=O)\C=C\c1ccc(O)c(O)c1 |r,wU:2.25,4.22,wD:1.0,7.8,4.4,(-1.06,-23.53,;-1.06,-21.99,;.29,-21.22,;.29,-19.66,;-1.06,-18.89,;.22,-18.03,;-2.39,-19.66,;-2.39,-21.22,;-3.73,-21.99,;-3.73,-23.53,;-2.39,-24.3,;-5.06,-24.3,;-5.06,-25.84,;-6.39,-26.61,;-7.73,-25.84,;-9.06,-26.61,;-9.06,-28.15,;-10.39,-28.92,;-7.72,-28.92,;-7.71,-30.46,;-6.39,-28.15,;-1.91,-17.6,;-.95,-16.4,;-3.45,-17.53,;1.62,-21.99,;1.61,-23.53,;.28,-24.3,;2.95,-24.3,;2.94,-25.84,;4.27,-26.62,;4.26,-28.16,;5.59,-28.93,;6.93,-28.16,;8.26,-28.93,;6.93,-26.61,;8.26,-25.84,;5.6,-25.85,)| Show InChI InChI=1S/C25H24O12/c26-15-5-1-13(9-17(15)28)3-7-21(30)36-19-11-25(35,24(33)34)12-20(23(19)32)37-22(31)8-4-14-2-6-16(27)18(29)10-14/h1-10,19-20,23,26-29,32,35H,11-12H2,(H,33,34)/b7-3+,8-4+/t19-,20-,23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Korean Institute of Oriental Medicine (KIOM)
Curated by ChEMBL
| Assay Description
Inhibition of Sprague-Dawley rat lens aldose reductase |
J Nat Prod 75: 267-70 (2012)
Article DOI: 10.1021/np200646e
BindingDB Entry DOI: 10.7270/Q2XS5WCH |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data