 Found 8 hits for monomerid = 50396073
Found 8 hits for monomerid = 50396073 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50396073
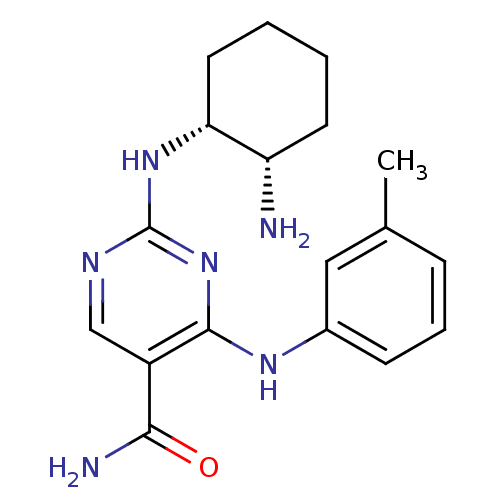
(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
MMDB
PDB
US Patent
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Portola Pharmaceuticals, Inc.
US Patent
| Assay Description
SYK tyrosine phosphorylation activity is measured using the LANCE Technology developed by Perkin Elmer Life and Analytical Sciences (Boston, Mass.). ... |
US Patent US8952027 (2015)
BindingDB Entry DOI: 10.7270/Q2CF9NV0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mitogen-activated protein kinase; ERK1/ERK2
(Homo sapiens (Human)) | BDBM50396073

(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D
Curated by ChEMBL
| Assay Description
Inhibition of Erk1/2 phosphorylation expressed in ramos cells after 30 mins by MSD assay |
Bioorg Med Chem Lett 21: 6188-94 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.082
BindingDB Entry DOI: 10.7270/Q2XD12ZW |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM50396073

(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant Flag-6His-Thr-tagged Aurora B assessed as phosphorylation of 5FAM-PKA-tide substrate after 150 mins by fuorescence po... |
Bioorg Med Chem Lett 21: 6188-94 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.082
BindingDB Entry DOI: 10.7270/Q2XD12ZW |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50396073

(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D
Curated by ChEMBL
| Assay Description
Inhibition of human ERG expressed in CHO-K1 cells after 2 hrs by Cy3b-Dofetilide-based fluorescence polarisation assay |
Bioorg Med Chem Lett 21: 6188-94 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.082
BindingDB Entry DOI: 10.7270/Q2XD12ZW |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50396073

(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rigel, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of SYK |
J Med Chem 55: 3614-43 (2012)
Article DOI: 10.1021/jm201271b
BindingDB Entry DOI: 10.7270/Q2NZ88RC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50396073

(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Rigel, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of SYK in human mast cell cultures assessed as reduction in Anti-IgG-stimulated FcgammaR-mediated leukotriene LTC4 production by fluoresce... |
J Med Chem 55: 3614-43 (2012)
Article DOI: 10.1021/jm201271b
BindingDB Entry DOI: 10.7270/Q2NZ88RC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50396073

(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D
Curated by ChEMBL
| Assay Description
Inhibition of human full-length recombinant 6His-SYK assessed as phosphorylation of Biotin-AAAEEIYGEI substrate after 60 mins by by TR-FRET assay |
Bioorg Med Chem Lett 21: 6188-94 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.082
BindingDB Entry DOI: 10.7270/Q2XD12ZW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50396073

(CHEMBL1235110)Show SMILES Cc1cccc(Nc2nc(N[C@@H]3CCCC[C@@H]3N)ncc2C(N)=O)c1 |r| Show InChI InChI=1S/C18H24N6O/c1-11-5-4-6-12(9-11)22-17-13(16(20)25)10-21-18(24-17)23-15-8-3-2-7-14(15)19/h4-6,9-10,14-15H,2-3,7-8,19H2,1H3,(H2,20,25)(H2,21,22,23,24)/t14-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 2.50E+3 | n/a | n/a | n/a | n/a |
Rigel, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of SYK in human platelet-rich plasma assessed as reduction in convulxin-induced GPV1/FcRgamma-mediated platelet aggregation |
J Med Chem 55: 3614-43 (2012)
Article DOI: 10.1021/jm201271b
BindingDB Entry DOI: 10.7270/Q2NZ88RC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data