

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50419256 CHEMBL1835065::US9579320, Example 302
SMILES: Cc1cccc(Nc2nc(N[C@@H]3CCOC[C@@H]3N)ncc2C(N)=O)c1
InChI Key: InChIKey=BDZSQIOBXDJLKM-UONOGXRCSA-N
Data: 5 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aurora kinase B (Homo sapiens (Human)) | BDBM50419256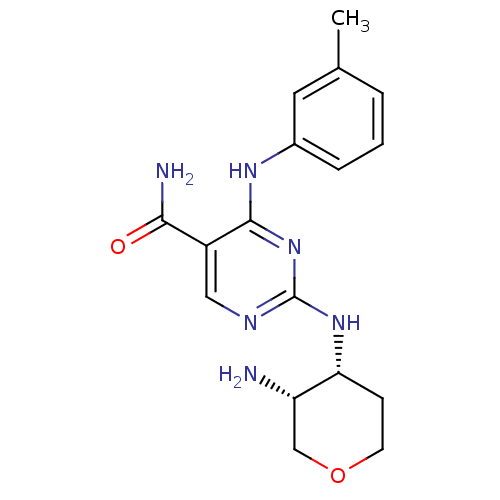 (CHEMBL1835065 | US9579320, Example 302) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.31E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D Curated by ChEMBL | Assay Description Inhibition of human recombinant Flag-6His-Thr-tagged Aurora B assessed as phosphorylation of 5FAM-PKA-tide substrate after 150 mins by fuorescence po... | Bioorg Med Chem Lett 21: 6188-94 (2011) Article DOI: 10.1016/j.bmcl.2011.07.082 BindingDB Entry DOI: 10.7270/Q2XD12ZW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase; ERK1/ERK2 (Homo sapiens (Human)) | BDBM50419256 (CHEMBL1835065 | US9579320, Example 302) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D Curated by ChEMBL | Assay Description Inhibition of Erk1/2 phosphorylation expressed in ramos cells after 30 mins by MSD assay | Bioorg Med Chem Lett 21: 6188-94 (2011) Article DOI: 10.1016/j.bmcl.2011.07.082 BindingDB Entry DOI: 10.7270/Q2XD12ZW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase SYK (Homo sapiens (Human)) | BDBM50419256 (CHEMBL1835065 | US9579320, Example 302) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 55 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Portola Pharmaceuticals, Inc. US Patent | Assay Description Potency of candidate molecules for inhibiting syk tyrosine phosphorylation activity is assessed by measuring the ability of a test compound to inhibi... | US Patent US9579320 (2017) BindingDB Entry DOI: 10.7270/Q2K07692 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50419256 (CHEMBL1835065 | US9579320, Example 302) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D Curated by ChEMBL | Assay Description Inhibition of human ERG expressed in CHO-K1 cells after 2 hrs by Cy3b-Dofetilide-based fluorescence polarisation assay | Bioorg Med Chem Lett 21: 6188-94 (2011) Article DOI: 10.1016/j.bmcl.2011.07.082 BindingDB Entry DOI: 10.7270/Q2XD12ZW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tyrosine-protein kinase SYK (Homo sapiens (Human)) | BDBM50419256 (CHEMBL1835065 | US9579320, Example 302) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline R&D Curated by ChEMBL | Assay Description Inhibition of human full-length recombinant 6His-SYK assessed as phosphorylation of Biotin-AAAEEIYGEI substrate after 60 mins by by TR-FRET assay | Bioorg Med Chem Lett 21: 6188-94 (2011) Article DOI: 10.1016/j.bmcl.2011.07.082 BindingDB Entry DOI: 10.7270/Q2XD12ZW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||