

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50438726 CHEMBL2414684
SMILES: Nc1ccnc2OCCN(c3ccc(cc3)[C@H]3CC[C@H](CC(O)=O)CC3)C(=O)c12
InChI Key: InChIKey=CJEWHTUUJIZSOG-SHTZXODSSA-N
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diacylglycerol O-acyltransferase 1 (DGAT1) (Homo sapiens (Human)) | BDBM50438726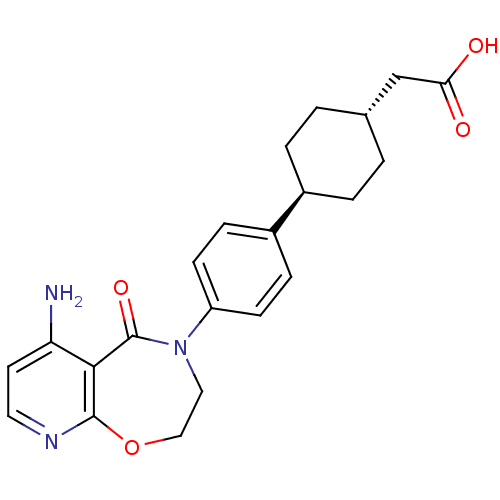 (CHEMBL2414684) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of human full length DGAT-1 expressed in insect sf9 cells using [14C]decanoylCoA as substrate after 1.5 hrs by scintillation spectrometry | Bioorg Med Chem 21: 5081-97 (2013) Article DOI: 10.1016/j.bmc.2013.06.045 BindingDB Entry DOI: 10.7270/Q26111RG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Diacylglycerol O-acyltransferase 1 (DGAT1) (Homo sapiens (Human)) | BDBM50438726 (CHEMBL2414684) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 99 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of DGAT-1-mediated triglyceride synthesis in human HT-29 cells using [3H]-glycerol as substrate incubated for 1 hr prior to substrate addi... | Bioorg Med Chem 21: 5081-97 (2013) Article DOI: 10.1016/j.bmc.2013.06.045 BindingDB Entry DOI: 10.7270/Q26111RG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||