

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560224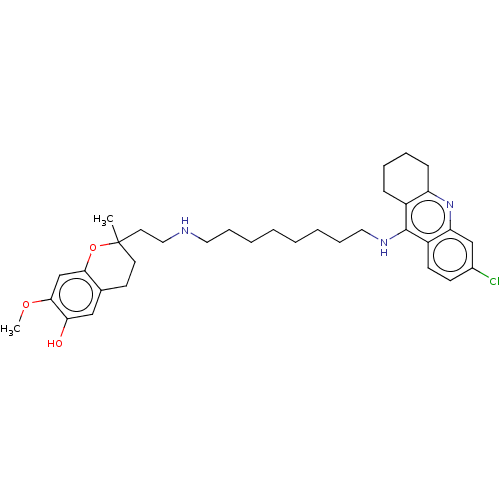 (CHEMBL4751100) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.132 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Mixed inhibition of recombinant human AChE expressed in HEK293 cells assessed as dissociation constant for enzyme-inhibitor complex using varying lev... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560224 (CHEMBL4751100) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.258 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Mixed inhibition of recombinant human AChE expressed in HEK293 cells assessed as dissociation constant for enzyme-substrate-inhibitor complex using v... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560224 (CHEMBL4751100) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560225 (CHEMBL4785201) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560223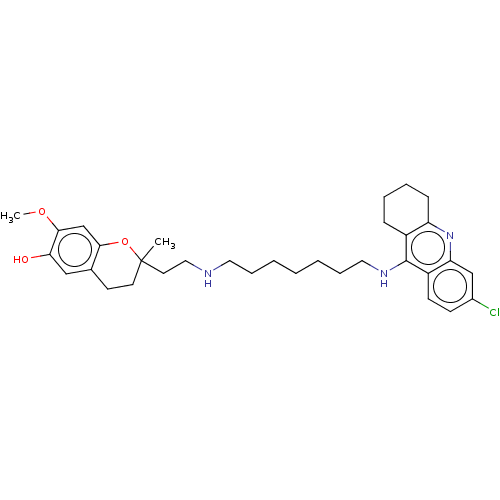 (CHEMBL4796461) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.440 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560218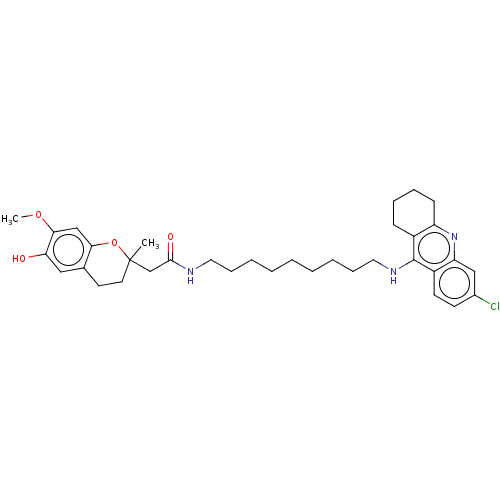 (CHEMBL4797814) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560217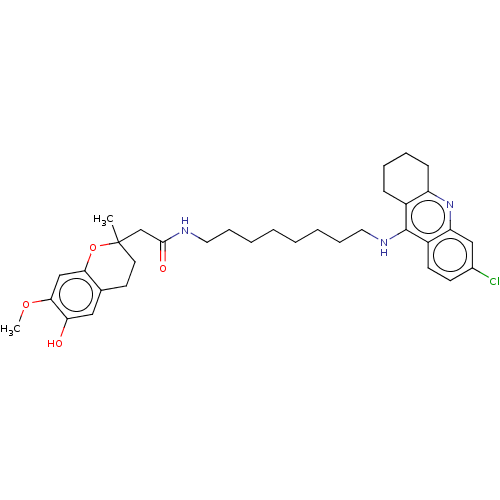 (CHEMBL4787579) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560220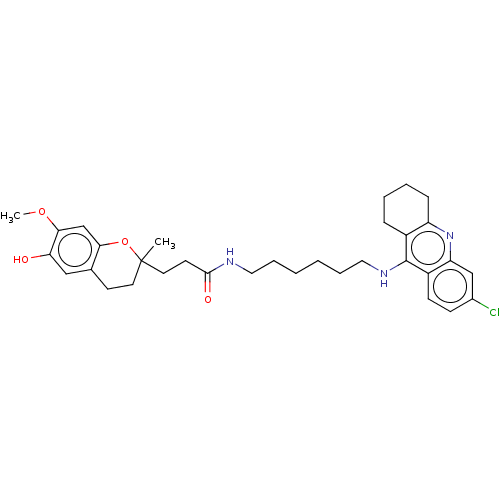 (CHEMBL4762419) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560212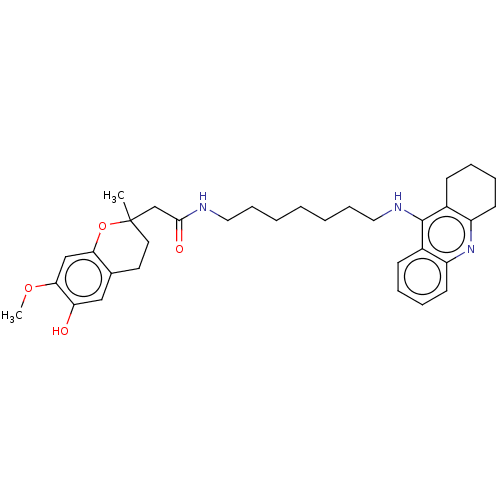 (CHEMBL4794549) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560209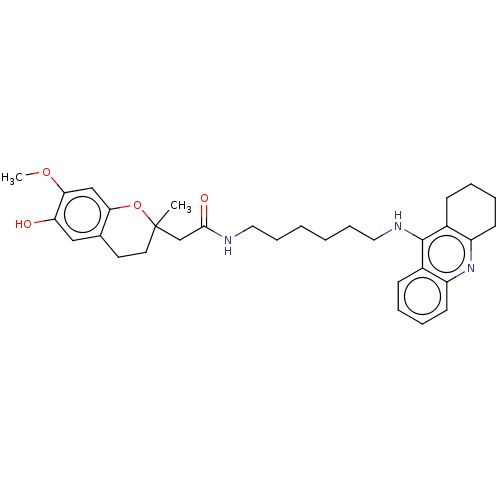 (CHEMBL4749077) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560211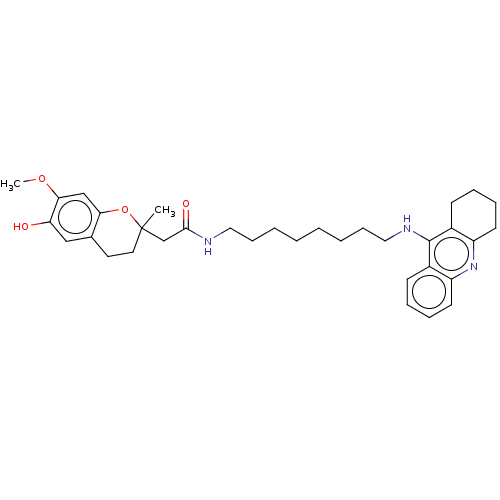 (CHEMBL4792460) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560216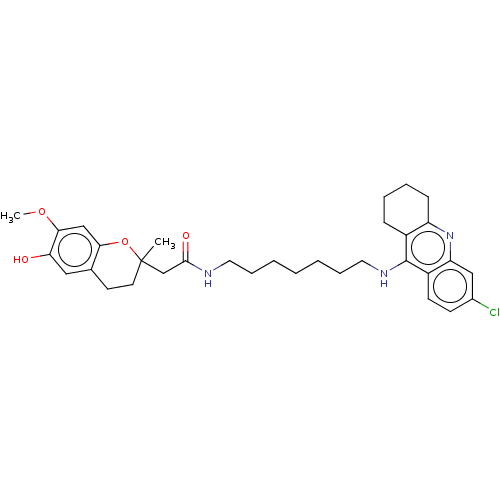 (CHEMBL4798535) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560210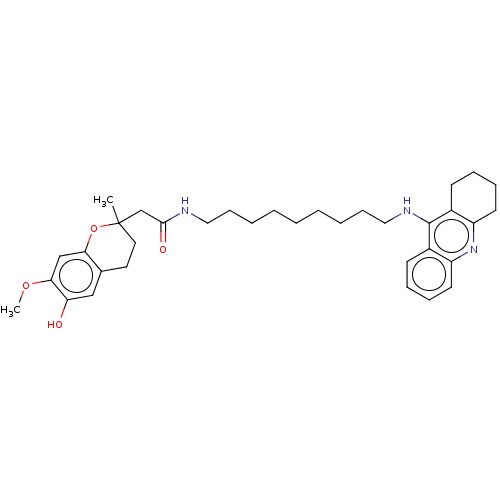 (CHEMBL4757922) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560222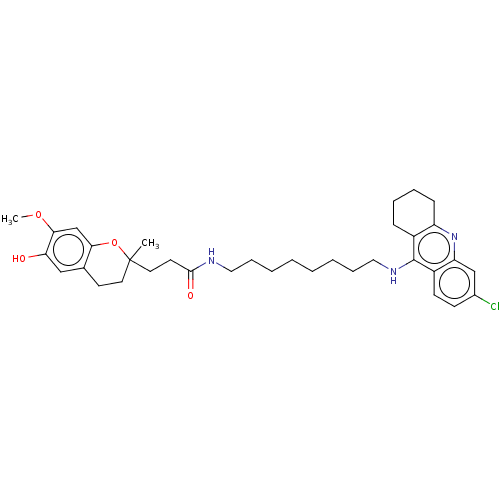 (CHEMBL4794645) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560227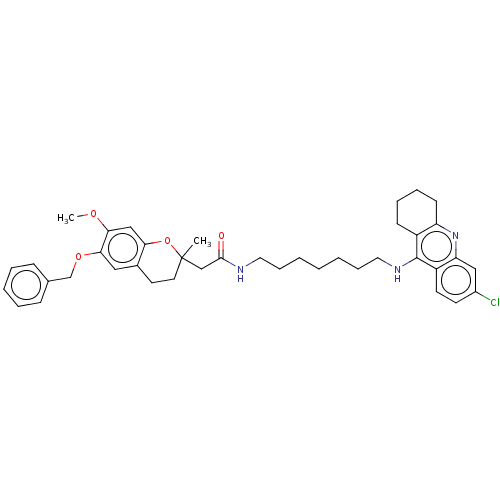 (CHEMBL4747041) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560214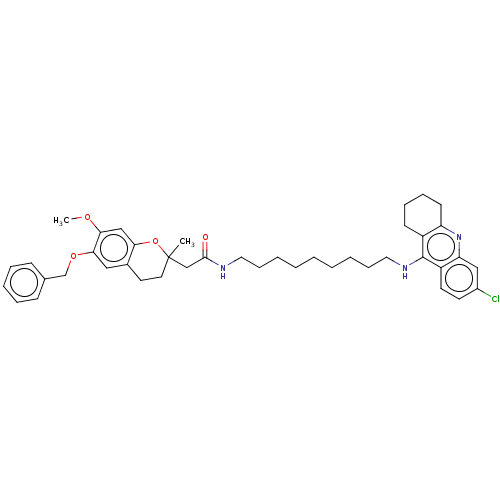 (CHEMBL4789620) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560221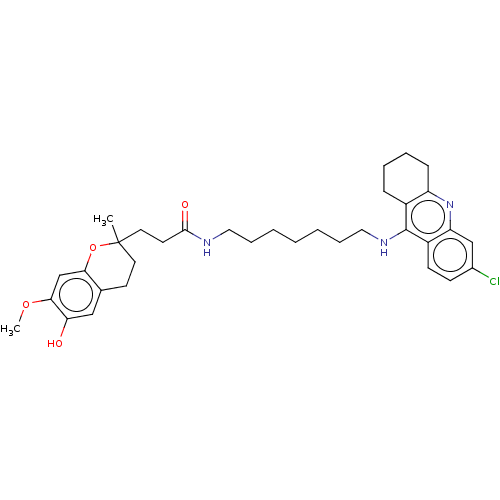 (CHEMBL4797177) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560226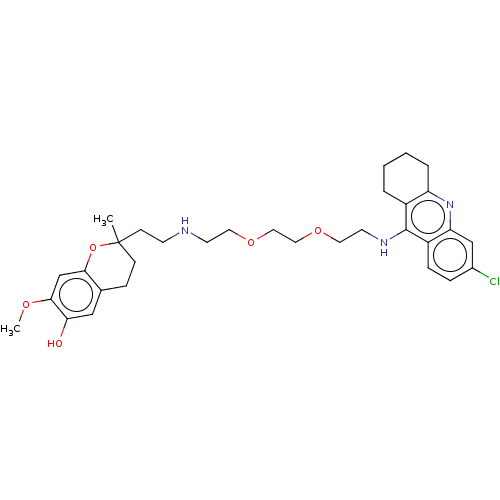 (CHEMBL4787251) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560215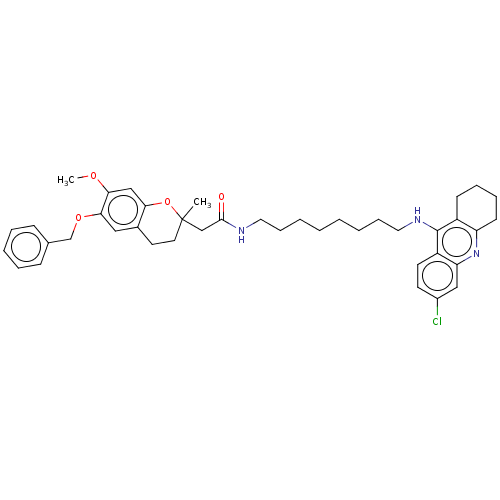 (CHEMBL4747836) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560213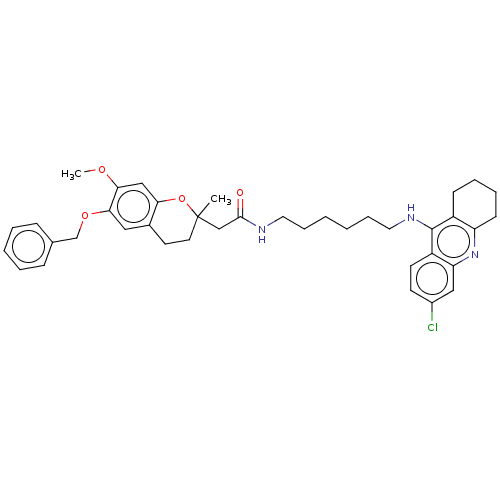 (CHEMBL4789172) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560224 (CHEMBL4751100) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8987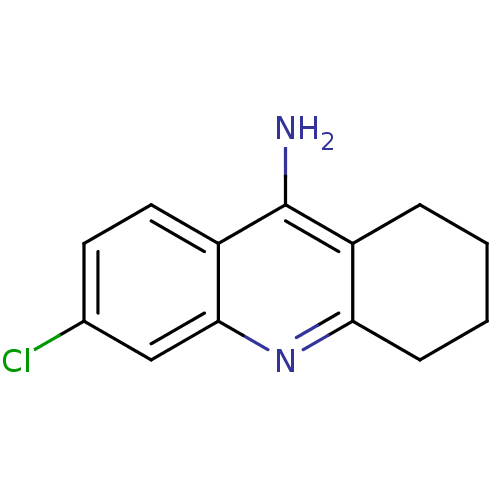 (6-chloro-1,2,3,4-tetrahydroacridin-9-amine | 6-chl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560223 (CHEMBL4796461) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560225 (CHEMBL4785201) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560214 (CHEMBL4789620) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560219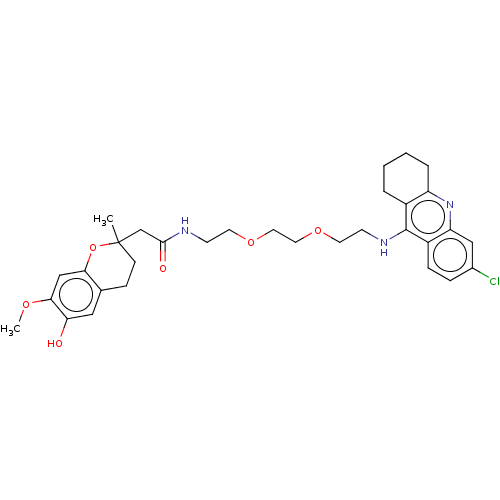 (CHEMBL4758624) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Rattus norvegicus (rat)) | BDBM50360256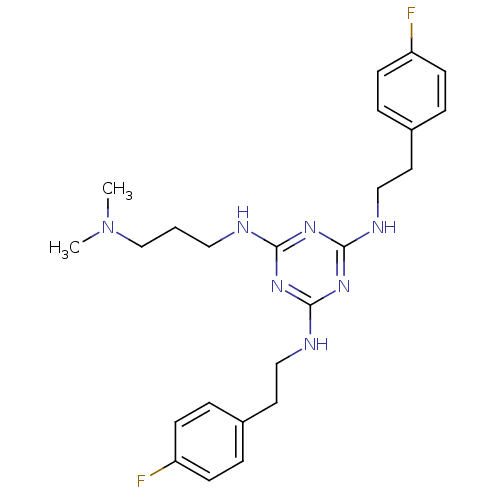 (CHEMBL1928201) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut de Qu£mica Avan£ada de Catalunya Curated by ChEMBL | Assay Description Antagonist activity at rat TRPV1 expressed in Xenopus laevis oocytes assessed as blockage of capsaicin-induced current at holding potential -60 mV by... | J Med Chem 54: 7441-52 (2011) Article DOI: 10.1021/jm200981s BindingDB Entry DOI: 10.7270/Q2M32W6F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560227 (CHEMBL4747041) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560210 (CHEMBL4757922) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 58 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560211 (CHEMBL4792460) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560216 (CHEMBL4798535) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 79 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Rattus norvegicus (rat)) | BDBM50360257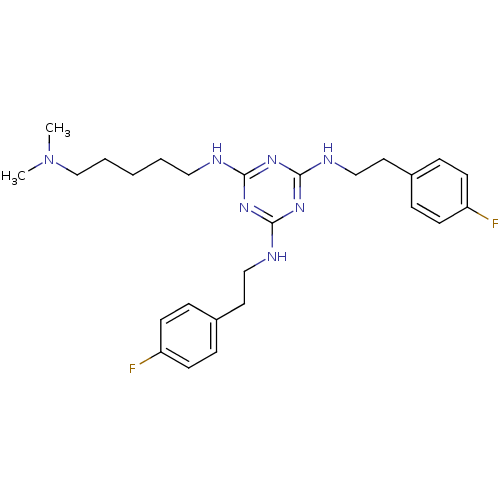 (CHEMBL1928202) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut de Qu£mica Avan£ada de Catalunya Curated by ChEMBL | Assay Description Antagonist activity at rat TRPV1 expressed in Xenopus laevis oocytes assessed as blockage of capsaicin-induced current at holding potential -60 mV by... | J Med Chem 54: 7441-52 (2011) Article DOI: 10.1021/jm200981s BindingDB Entry DOI: 10.7270/Q2M32W6F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560215 (CHEMBL4747836) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560220 (CHEMBL4762419) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 102 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Squalene monooxygenase (Rattus norvegicus) | BDBM50281098 (2,2-dimethyl-3-[3,7,12-trimethyl-14-[3-methyl-3-(4...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for its inhibitory activity against squalene epoxidase from rat liver microsomes | Bioorg Med Chem Lett 3: 2581-2586 (1993) Article DOI: 10.1016/S0960-894X(01)80720-4 BindingDB Entry DOI: 10.7270/Q24F1QNX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lanosterol synthase (Rattus norvegicus) | BDBM50280127 (2,2-dimethyl-3-[3,7,12-trimethyl-14-[3-methyl-3-(4...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for inhibitory activity against 2,3-oxidosqualene-lanosterol cyclase in rat liver microsomes | Bioorg Med Chem Lett 2: 1239-1242 (1992) Article DOI: 10.1016/S0960-894X(00)80221-8 BindingDB Entry DOI: 10.7270/Q28K791S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560212 (CHEMBL4794549) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 111 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560222 (CHEMBL4794645) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 148 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560218 (CHEMBL4797814) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 168 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560217 (CHEMBL4787579) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560226 (CHEMBL4787251) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560221 (CHEMBL4797177) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 187 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50560213 (CHEMBL4789172) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 201 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50560209 (CHEMBL4749077) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 275 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Rattus norvegicus (rat)) | BDBM50360242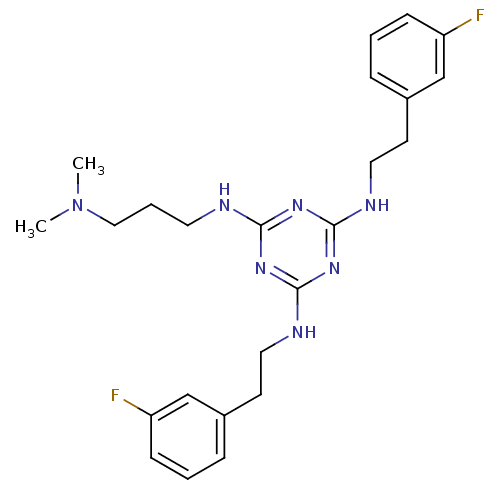 (CHEMBL1928218) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut de Qu£mica Avan£ada de Catalunya Curated by ChEMBL | Assay Description Antagonist activity at rat TRPV1 expressed in Xenopus laevis oocytes assessed as blockage of capsaicin-induced current at holding potential -60 mV by... | J Med Chem 54: 7441-52 (2011) Article DOI: 10.1021/jm200981s BindingDB Entry DOI: 10.7270/Q2M32W6F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM8961 (1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human AChE expressed in HEK293 cells using acetylthiocholine iodide as substrate preincubated for 15 mins followed by subst... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Rattus norvegicus (rat)) | BDBM50360241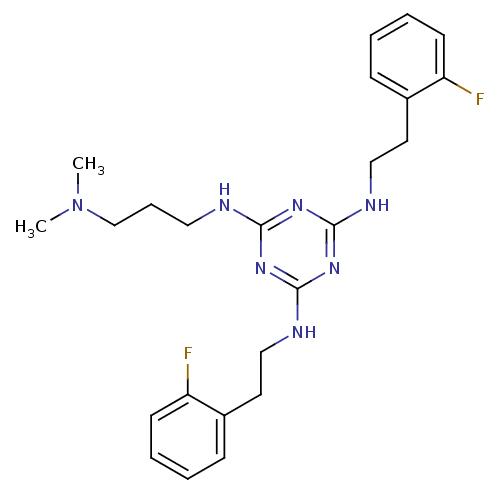 (CHEMBL1928217) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 450 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut de Qu£mica Avan£ada de Catalunya Curated by ChEMBL | Assay Description Antagonist activity at rat TRPV1 expressed in Xenopus laevis oocytes assessed as blockage of capsaicin-induced current at holding potential -60 mV by... | J Med Chem 54: 7441-52 (2011) Article DOI: 10.1021/jm200981s BindingDB Entry DOI: 10.7270/Q2M32W6F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Rattus norvegicus (rat)) | BDBM50360263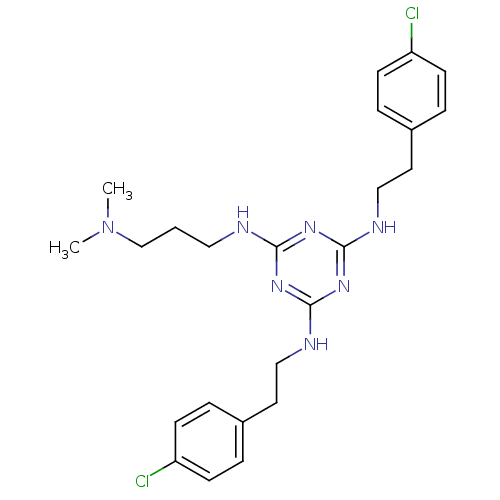 (CHEMBL1928208) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Institut de Qu£mica Avan£ada de Catalunya Curated by ChEMBL | Assay Description Antagonist activity at rat TRPV1 expressed in Xenopus laevis oocytes assessed as blockage of capsaicin-induced current at holding potential -60 mV by... | J Med Chem 54: 7441-52 (2011) Article DOI: 10.1021/jm200981s BindingDB Entry DOI: 10.7270/Q2M32W6F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM8987 (6-chloro-1,2,3,4-tetrahydroacridin-9-amine | 6-chl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 505 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 15 mins followed by substrate addition by Ellman's... | Citation and Details Article DOI: 10.1021/acs.jmedchem.0c00528 BindingDB Entry DOI: 10.7270/Q23N2733 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 97 total ) | Next | Last >> |