 Found 2072 hits with Last Name = 'kahn' and Initial = 'd'
Found 2072 hits with Last Name = 'kahn' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Proto-oncogene tyrosine-protein kinase receptor Ret [G810S]
(Homo sapiens (Human)) | BDBM576988
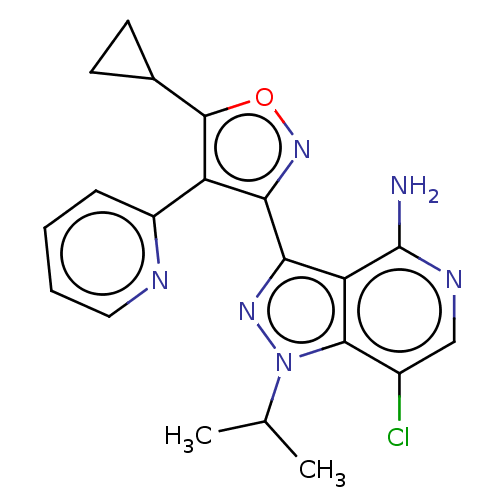
(US11472802, Example 8)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617349

(US20230279025, Example 465)Show SMILES CCc1cccc2cccc(-c3ncc4c(nc(OC[C@@]56CCCN5C[C@H](F)C6)nc4c3F)N3CC4CCC(C3)N4)c12 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617355
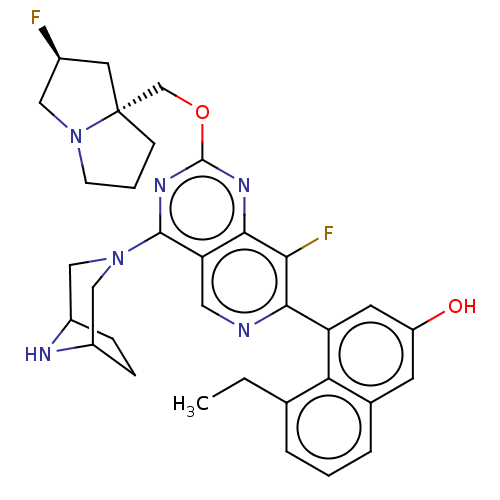
(US20230279025, Example 468)Show SMILES CCc1cccc2cc(O)cc(-c3ncc4c(nc(OC[C@]56CCCN5C[C@@H](F)C6)nc4c3F)N3CC4CCC(C3)N4)c12 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM50579601
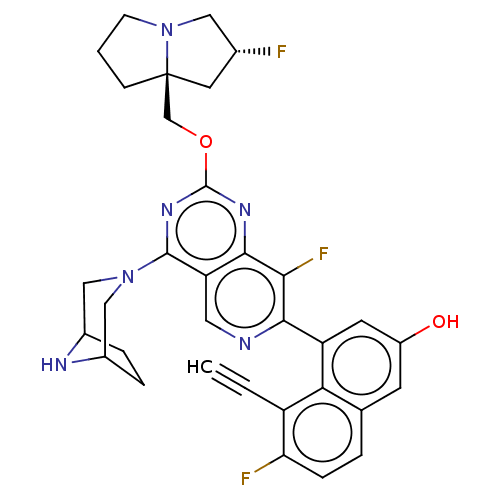
(CHEMBL4858364 | US11453683, Example 252 | US202302...)Show SMILES Oc1cc(-c2ncc3c(nc(OC[C@@]45CCCN4C[C@H](F)C5)nc3c2F)N2CC3CCC(C2)N3)c2c(C#C)c(F)ccc2c1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| | n/a | n/a | <0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617323
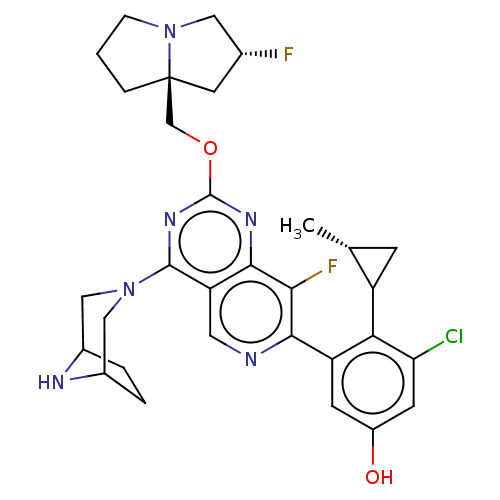
(US20230279025, Example 452 | US20230279025, Exampl...)Show SMILES C[C@@H]1CC1c1c(Cl)cc(O)cc1-c1ncc2c(nc(OC[C@@]34CCCN3C[C@H](F)C4)nc2c1F)N1CC2CCC(C1)N2 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617363

(US20230279025, Example 482)Show SMILES Oc1cc(-c2ncc3c(nc(OC[C@]45CCCN4C[C@@H](F)C5)nc3c2F)N2CC3CCC(C2)N3)c2c(C#C)c(F)ccc2c1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617357

(US20230279025, Example 469)Show SMILES Oc1cc(-c2ncc3c(nc(OC[C@]45C[C@@H](F)CN4CCCC5)nc3c2F)N2CC3CCC(C2)N3)c2c(C#C)c(F)ccc2c1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM573400

(US11453683, Example 259 | US20230279025, Example 4...)Show SMILES CCc1cccc2cc(O)cc(-c3ncc4c(nc(OC[C@@]56CCCN5C[C@H](F)C6)nc4c3F)N3CC4CCC(C3)N4)c12 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <0.00200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617351
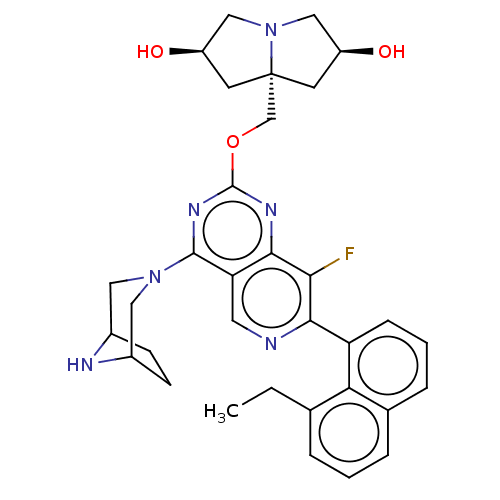
(US20230279025, Example 466)Show SMILES CCc1cccc2cccc(-c3ncc4c(nc(OC[C@]56C[C@@H](O)CN5C[C@@H](O)C6)nc4c3F)N3CC4CCC(C3)N4)c12 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617347
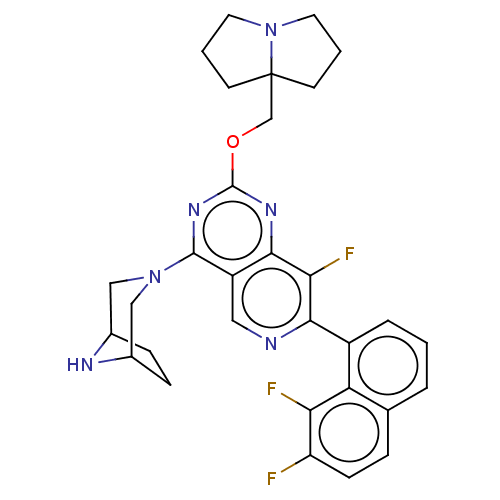
(US20230279025, Example 464)Show SMILES Fc1ccc2cccc(-c3ncc4c(nc(OCC56CCCN5CCC6)nc4c3F)N3CC4CCC(C3)N4)c2c1F | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617345
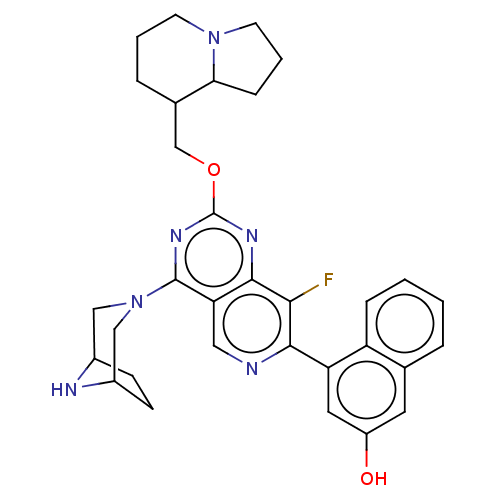
(US20230279025, Example 463)Show SMILES Oc1cc(-c2ncc3c(nc(OCC4CCCN5CCCC45)nc3c2F)N2CC3CCC(C2)N3)c2ccccc2c1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [V804M]
(Homo sapiens (Human)) | BDBM576988

(US11472802, Example 8)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM576988

(US11472802, Example 8)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM576988

(US11472802, Example 8)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM617339

(US20230279025, Example 460)Show SMILES Fc1ccc2cccc(-c3ncc4c(nc(OCC56CCCN5CCC6)nc4c3F)N3CC4CCC(C3)N4)c2c1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM616938
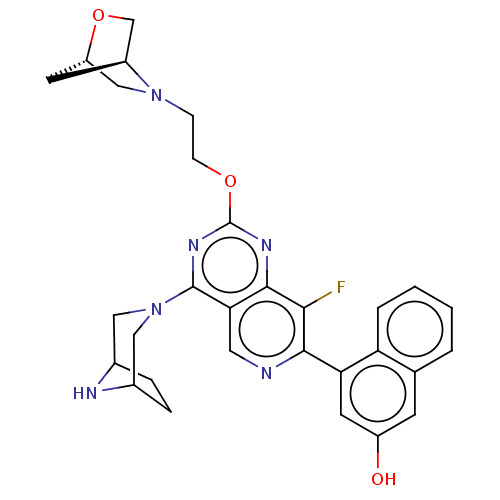
(US20230279025, Example 26 | US20230279025, Example...)Show SMILES Oc1cc(-c2ncc3c(nc(OCCN4C[C@@H]5C[C@H]4CO5)nc3c2F)N2CC3CCC(C2)N3)c2ccccc2c1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM573426

(US11453683, Example 284 | US20230279025, Example 2...)Show SMILES Oc1cc(Cl)c(c(c1)-c1ncc2c(nc(OC[C@@]34CCCN3C[C@H](F)C4)nc2c1F)N1CC2CCC(C1)N2)C(F)(F)F |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577053

(US11472802, Example 58)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2c[nH]cn2)c2c(N)nccc12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [G810R]
(Homo sapiens (Human)) | BDBM576988

(US11472802, Example 8)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM573426

(US11453683, Example 284 | US20230279025, Example 2...)Show SMILES Oc1cc(Cl)c(c(c1)-c1ncc2c(nc(OC[C@@]34CCCN3C[C@H](F)C4)nc2c1F)N1CC2CCC(C1)N2)C(F)(F)F |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Table 2: The ability of a compound to bind to KRAS G12D was measured using a TR-FRET displacement assay. Biotinylated GDP-loaded recombinant human KR... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2PR807K |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577053

(US11472802, Example 58)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2c[nH]cn2)c2c(N)nccc12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577049

(US11472802, Example 55)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577049

(US11472802, Example 55)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577050

(7-chloro-3-(5- cyclopropyl-4-(1H- imidazol-4- yl)i...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2c[nH]cn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM50579600
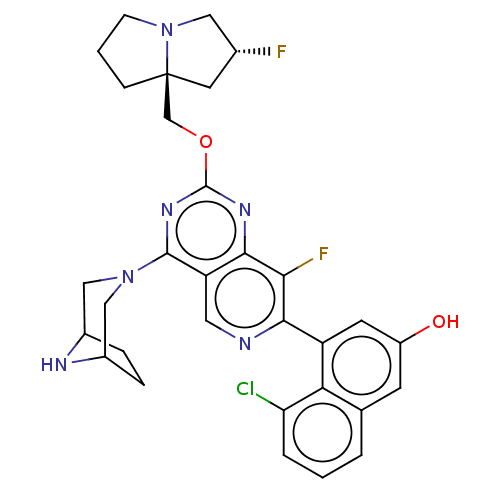
(CHEMBL4857438 | US11453683, Example 251 | US202302...)Show SMILES Oc1cc(-c2ncc3c(nc(OC[C@@]45CCCN4C[C@H](F)C5)nc3c2F)N2CC3CCC(C2)N3)c2c(Cl)cccc2c1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Table 2: The ability of a compound to bind to KRAS G12D was measured using a TR-FRET displacement assay. Biotinylated GDP-loaded recombinant human KR... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2PR807K |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577046
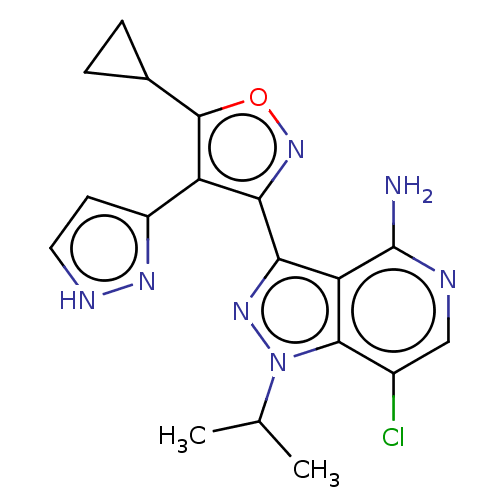
(7-chloro-3-(5-cyclopropyl- 4-(1H-pyrazol-3- yl)iso...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc[nH]n2)c2c(N)ncc(Cl)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577046

(7-chloro-3-(5-cyclopropyl- 4-(1H-pyrazol-3- yl)iso...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc[nH]n2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM50579600

(CHEMBL4857438 | US11453683, Example 251 | US202302...)Show SMILES Oc1cc(-c2ncc3c(nc(OC[C@@]45CCCN4C[C@H](F)C5)nc3c2F)N2CC3CCC(C2)N3)c2c(Cl)cccc2c1 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577063
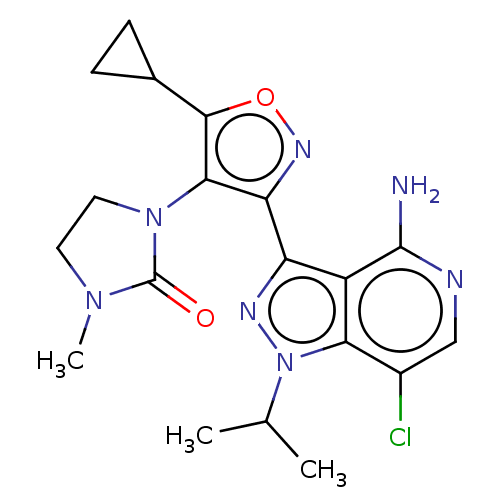
(1-(3-(4-amino-7-chloro-1- isopropyl-1H-pyrazolo[4,...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCN(C)C2=O)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [V804M]
(Homo sapiens (Human)) | BDBM577049

(US11472802, Example 55)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM576996

(US11472802, Example 11)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(F)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM50579595

(CHEMBL4863339 | US11453683, Example 185 | US202302...)Show SMILES Oc1cc(-c2ncc3c(nc(OCC45CCCN4CCC5)nc3c2F)N2CC3CCC(C2)N3)c2c(cccc2c1)C#C | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577061

(US11472802, Example 65)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCC2=O)c2c(N)ncc(Cl)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577061

(US11472802, Example 65)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCC2=O)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577063

(1-(3-(4-amino-7-chloro-1- isopropyl-1H-pyrazolo[4,...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2N2CCN(C)C2=O)c2c(N)ncc(Cl)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577064
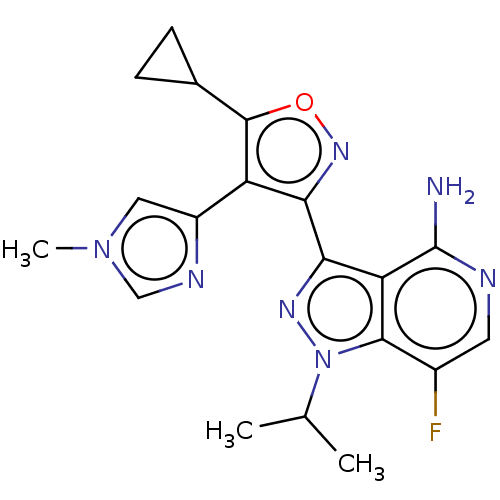
(US11472802, Example 68)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)ncc(F)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577064

(US11472802, Example 68)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cn(C)cn2)c2c(N)ncc(F)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577066
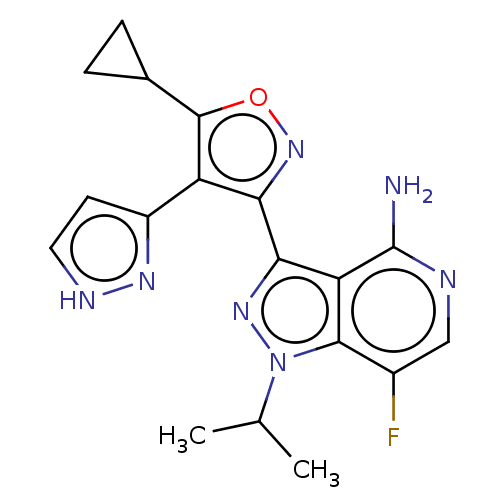
(3-(5-cyclopropyl- 4-(1H-pyrazol-3- yl)isoxazol-3- ...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc[nH]n2)c2c(N)ncc(F)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577025
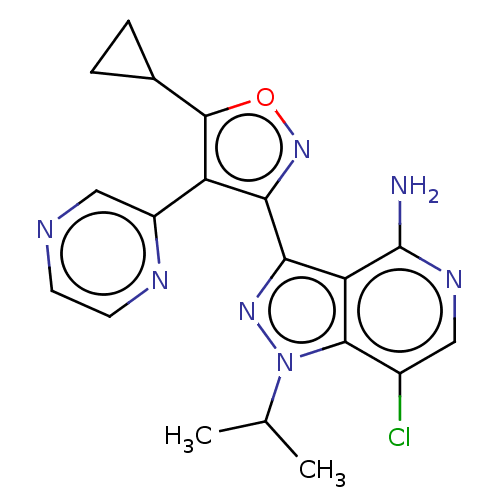
(7-chloro-3-(5-cyclopropyl-4- (pyrazin-2-yl)isoxazo...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cnccn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577026

(7-chloro-3-(5-cyclopropyl-4-(4- methylpyridin-2-yl...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc(C)ccn2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [M918T]
(Homo sapiens (Human)) | BDBM577045

(US11472802, Example 51)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc(C)[nH]n2)c2c(N)ncc(Cl)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM50579595

(CHEMBL4863339 | US11453683, Example 185 | US202302...)Show SMILES Oc1cc(-c2ncc3c(nc(OCC45CCCN4CCC5)nc3c2F)N2CC3CCC(C2)N3)c2c(cccc2c1)C#C | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Table 2: The ability of a compound to bind to KRAS G12D was measured using a TR-FRET displacement assay. Biotinylated GDP-loaded recombinant human KR... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2PR807K |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM576996

(US11472802, Example 11)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(F)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577050

(7-chloro-3-(5- cyclopropyl-4-(1H- imidazol-4- yl)i...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2c[nH]cn2)c2c(N)ncc(Cl)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf [V600E]
(Homo sapiens (Human)) | BDBM565148

(US11414404, Ex. # 25 | US11634409, Example 25)Show SMILES Cc1c(Nc2c(F)ccc(NS(=O)(=O)c3cc(F)ccc3F)c2Cl)ccc2ncn(C)c(=O)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
A competitive displacement assay was configured for B-Raf that monitors the amount of a fluorescently-tagged �tracer� bound to B-Raf via TR-FRET from... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2JD511G |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf [V600E]
(Homo sapiens (Human)) | BDBM565148

(US11414404, Ex. # 25 | US11634409, Example 25)Show SMILES Cc1c(Nc2c(F)ccc(NS(=O)(=O)c3cc(F)ccc3F)c2Cl)ccc2ncn(C)c(=O)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2FT8R02 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM577067
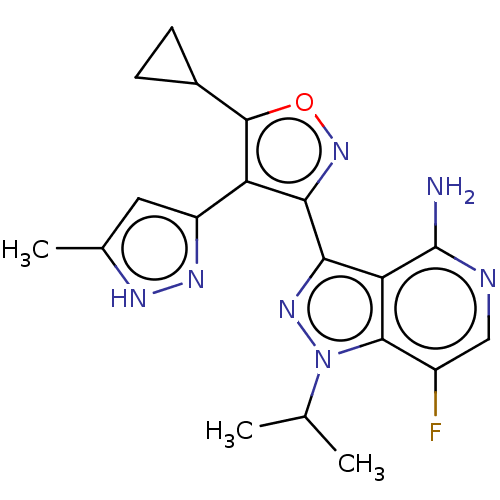
(3-(5-cyclopropyl- 4-(5-methyl-1H- pyrazol-3-yl) is...)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2cc(C)[nH]n2)c2c(N)ncc(F)c12 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The potency of compounds inhibiting several different RET kinase forms (Wild Type, V804M, M918T, G810R, & G810S) were determined using CisBio's H... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret [V804M]
(Homo sapiens (Human)) | BDBM576996

(US11472802, Example 11)Show SMILES CC(C)n1nc(-c2noc(C3CC3)c2-c2ccccn2)c2c(N)ncc(F)c12 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q22N55HG |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM573644

(US11453683, Example 439 | US20230279025, Example 4...)Show SMILES CN(C)C(=O)OC[C@H]1CC[C@@]2(COc3nc(N4CC5CCC(C4)N5)c4cnc(c(F)c4n3)-c3cccc4ccc(F)c(C#C)c34)CCCN12 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |
GTPase KRas [G12D]
(Homo sapiens (Human)) | BDBM573433
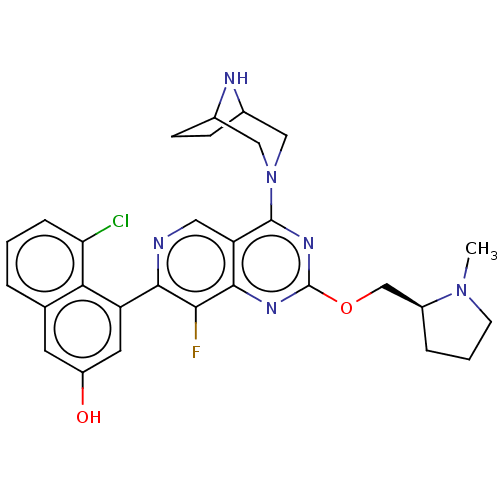
(US11453683, Example 63 | US20230279025, Example 63)Show SMILES CN1CCC[C@H]1COc1nc(N2CC3CCC(C2)N3)c2cnc(c(F)c2n1)-c1cc(O)cc2cccc(Cl)c12 |r| | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2QR5281 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data