 Found 528 hits with Last Name = 'landi' and Initial = 'g'
Found 528 hits with Last Name = 'landi' and Initial = 'g' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600683
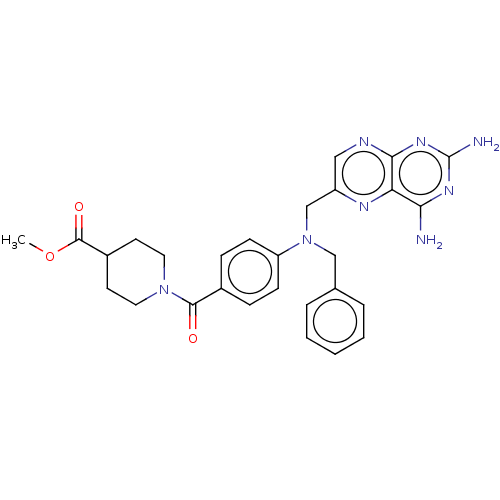
(CHEMBL5184936)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(Cc1ccccc1)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600703
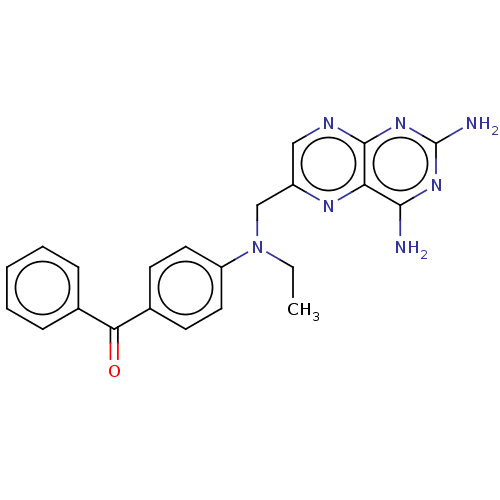
(CHEMBL5201183)Show SMILES CCN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)c1ccccc1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600694

(CHEMBL5204239)Show SMILES Nc1nc(N)c2nc(CNc3ccc(cc3)C(=O)NCCc3ccccc3)cnc2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000806

(13-[2-Amino-3-(4-hydroxy-2-methyl-phenyl)-propiony...)Show SMILES Cc1cc(O)ccc1C[C@H](N)C(=O)N[C@H]1C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](C(O)=O)C(C)(C)SSC1(C)C Show InChI InChI=1S/C31H41N5O7S2/c1-17-13-20(37)12-11-19(17)15-21(32)26(39)35-24-28(41)33-16-23(38)34-22(14-18-9-7-6-8-10-18)27(40)36-25(29(42)43)31(4,5)45-44-30(24,2)3/h6-13,21-22,24-25,37H,14-16,32H2,1-5H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t21-,22-,24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.890 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]- DPDPE (opioid receptor delta selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600702
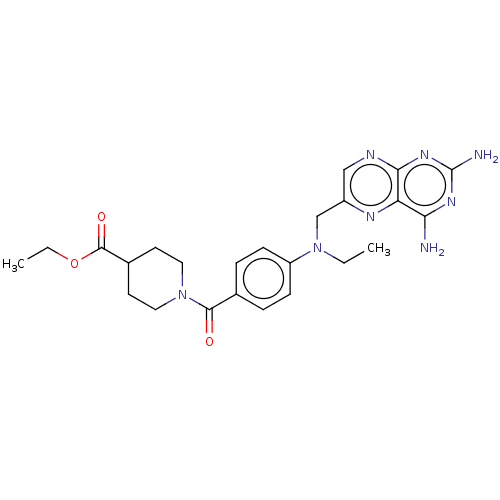
(CHEMBL5199171)Show SMILES CCOC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(CC)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600693

(CHEMBL5175012)Show SMILES Nc1nc(N)c2nc(CNc3ccc(cc3)C(=O)c3ccccc3)cnc2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600684

(CHEMBL5182125)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(SCc2cnc3nc(N)nc(N)c3n2)nc1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50001683

(13-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-7...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)cc2)C(=O)NCC(=O)N[C@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O Show InChI InChI=1S/C30H39N5O7S2/c1-29(2)23(34-25(38)20(31)14-18-10-12-19(36)13-11-18)27(40)32-16-22(37)33-21(15-17-8-6-5-7-9-17)26(39)35-24(28(41)42)30(3,4)44-43-29/h5-13,20-21,23-24,36H,14-16,31H2,1-4H3,(H,32,40)(H,33,37)(H,34,38)(H,35,39)(H,41,42)/t20-,21+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]-[p-Cl-Phe4]-DPDPE (opioid receptor delta selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000805

(13-[(2-Amino-6-hydroxy-1,2,3,4,4a,8a-hexahydro-nap...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@]2(N)CCC3CC(=O)C=CC3C2)C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O |c:20| Show InChI InChI=1S/C32H43N5O7S2/c1-30(2)24(37-29(44)32(33)13-12-19-15-21(38)11-10-20(19)16-32)27(41)34-17-23(39)35-22(14-18-8-6-5-7-9-18)26(40)36-25(28(42)43)31(3,4)46-45-30/h5-11,19-20,22,24-25H,12-17,33H2,1-4H3,(H,34,41)(H,35,39)(H,36,40)(H,37,44)(H,42,43)/t19?,20?,22-,24-,25-,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 2.36 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]-[p-Cl-Phe4]-DPDPE (opioid receptor delta selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50001683

(13-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-7...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)cc2)C(=O)NCC(=O)N[C@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O Show InChI InChI=1S/C30H39N5O7S2/c1-29(2)23(34-25(38)20(31)14-18-10-12-19(36)13-11-18)27(40)32-16-22(37)33-21(15-17-8-6-5-7-9-17)26(39)35-24(28(41)42)30(3,4)44-43-29/h5-13,20-21,23-24,36H,14-16,31H2,1-4H3,(H,32,40)(H,33,37)(H,34,38)(H,35,39)(H,41,42)/t20-,21+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Binding potency of compound in competition with [3H]DPDPE for Opioid receptor delta 1 tested in rat brain membrane |
J Med Chem 32: 638-43 (1989)
BindingDB Entry DOI: 10.7270/Q2DN45N0 |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600701
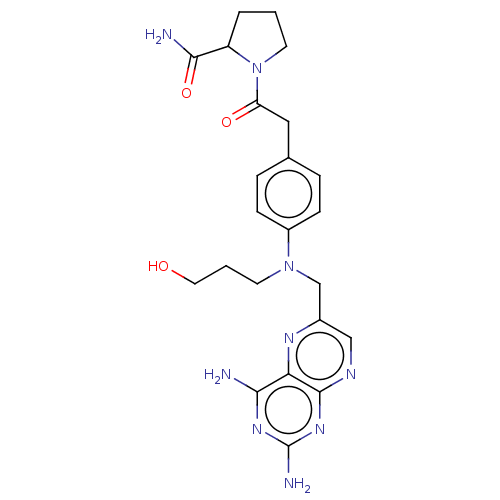
(CHEMBL5200612)Show SMILES NC(=O)C1CCCN1C(=O)Cc1ccc(cc1)N(CCCO)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50001683

(13-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-7...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)cc2)C(=O)NCC(=O)N[C@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O Show InChI InChI=1S/C30H39N5O7S2/c1-29(2)23(34-25(38)20(31)14-18-10-12-19(36)13-11-18)27(40)32-16-22(37)33-21(15-17-8-6-5-7-9-17)26(39)35-24(28(41)42)30(3,4)44-43-29/h5-13,20-21,23-24,36H,14-16,31H2,1-4H3,(H,32,40)(H,33,37)(H,34,38)(H,35,39)(H,41,42)/t20-,21+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for its inhibitory potency against opioid receptor delta of Mouse vas deferens |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50000806

(13-[2-Amino-3-(4-hydroxy-2-methyl-phenyl)-propiony...)Show SMILES Cc1cc(O)ccc1C[C@H](N)C(=O)N[C@H]1C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](C(O)=O)C(C)(C)SSC1(C)C Show InChI InChI=1S/C31H41N5O7S2/c1-17-13-20(37)12-11-19(17)15-21(32)26(39)35-24-28(41)33-16-23(38)34-22(14-18-9-7-6-8-10-18)27(40)36-25(29(42)43)31(4,5)45-44-30(24,2)3/h6-13,21-22,24-25,37H,14-16,32H2,1-5H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t21-,22-,24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for its inhibitory potency against opioid receptor delta of Mouse vas deferens |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50001683

(13-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-7...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)cc2)C(=O)NCC(=O)N[C@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O Show InChI InChI=1S/C30H39N5O7S2/c1-29(2)23(34-25(38)20(31)14-18-10-12-19(36)13-11-18)27(40)32-16-22(37)33-21(15-17-8-6-5-7-9-17)26(39)35-24(28(41)42)30(3,4)44-43-29/h5-13,20-21,23-24,36H,14-16,31H2,1-4H3,(H,32,40)(H,33,37)(H,34,38)(H,35,39)(H,41,42)/t20-,21+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]- DPDPE (opioid receptor delta selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600679

(CHEMBL426 | Methotrexate | TCMDC-123832 | TCMDC-12...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase 1
(Leishmania major) | BDBM50050426

(6-(Naphthalen-1-ylaminomethyl)-pteridine-2,4-diami...)Show InChI InChI=1S/C17H15N7/c18-15-14-16(24-17(19)23-15)21-9-11(22-14)8-20-13-7-3-5-10-4-1-2-6-12(10)13/h1-7,9,20H,8H2,(H4,18,19,21,23,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600700

(CHEMBL5188304)Show SMILES NC(=O)C1CCN(CC1)C(=O)Cc1ccc(cc1)N(CCCO)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600692

(CHEMBL5197107)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N1CCCCC1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600689
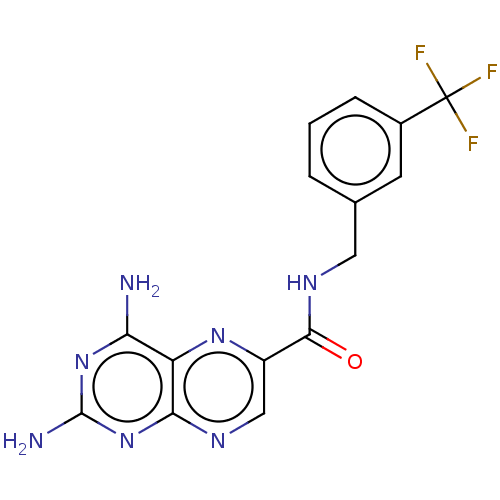
(CHEMBL5175397)Show SMILES Nc1nc(N)c2nc(cnc2n1)C(=O)NCc1cccc(c1)C(F)(F)F | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600688
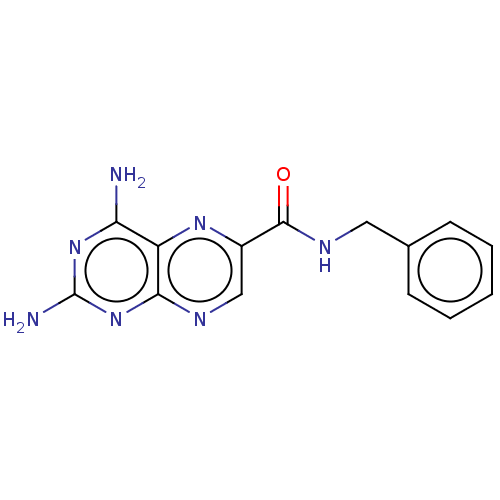
(CHEMBL5198855) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50050426

(6-(Naphthalen-1-ylaminomethyl)-pteridine-2,4-diami...)Show InChI InChI=1S/C17H15N7/c18-15-14-16(24-17(19)23-15)21-9-11(22-14)8-20-13-7-3-5-10-4-1-2-6-12(10)13/h1-7,9,20H,8H2,(H4,18,19,21,23,24) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50000799

(13-[2-Amino-3-(4-hydroxy-phenyl)-butyrylamino]-7-b...)Show SMILES C[C@@H]([C@H](N)C(=O)N[C@H]1C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](C(O)=O)C(C)(C)SSC1(C)C)c1ccc(O)cc1 Show InChI InChI=1S/C31H41N5O7S2/c1-17(19-11-13-20(37)14-12-19)23(32)27(40)35-24-28(41)33-16-22(38)34-21(15-18-9-7-6-8-10-18)26(39)36-25(29(42)43)31(4,5)45-44-30(24,2)3/h6-14,17,21,23-25,37H,15-16,32H2,1-5H3,(H,33,41)(H,34,38)(H,35,40)(H,36,39)(H,42,43)/t17-,21+,23+,24+,25+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the percentage of maximum inhibition at Opioid receptor delta 1 |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase 1
(Leishmania major) | BDBM50600683

(CHEMBL5184936)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(Cc1ccccc1)Cc1cnc2nc(N)nc(N)c2n1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000807

(13-[2-Amino-3-(4-hydroxy-3-methoxy-phenyl)-propion...)Show SMILES COc1cc(C[C@H](N)C(=O)N[C@H]2C(=O)NCC(=O)N[C@@H](Cc3ccccc3)C(=O)N[C@@H](C(O)=O)C(C)(C)SSC2(C)C)ccc1O Show InChI InChI=1S/C31H41N5O8S2/c1-30(2)24(35-26(39)19(32)13-18-11-12-21(37)22(15-18)44-5)28(41)33-16-23(38)34-20(14-17-9-7-6-8-10-17)27(40)36-25(29(42)43)31(3,4)46-45-30/h6-12,15,19-20,24-25,37H,13-14,16,32H2,1-5H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t19-,20-,24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]- DPDPE (delta opioid receptor selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50000805

(13-[(2-Amino-6-hydroxy-1,2,3,4,4a,8a-hexahydro-nap...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@]2(N)CCC3CC(=O)C=CC3C2)C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O |c:20| Show InChI InChI=1S/C32H43N5O7S2/c1-30(2)24(37-29(44)32(33)13-12-19-15-21(38)11-10-20(19)16-32)27(41)34-17-23(39)35-22(14-18-8-6-5-7-9-18)26(40)36-25(28(42)43)31(3,4)46-45-30/h5-11,19-20,22,24-25H,12-17,33H2,1-4H3,(H,34,41)(H,35,39)(H,36,40)(H,37,44)(H,42,43)/t19?,20?,22-,24-,25-,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for its inhibitory potency against opioid receptor delta of Mouse vas deferens |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000805

(13-[(2-Amino-6-hydroxy-1,2,3,4,4a,8a-hexahydro-nap...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@]2(N)CCC3CC(=O)C=CC3C2)C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O |c:20| Show InChI InChI=1S/C32H43N5O7S2/c1-30(2)24(37-29(44)32(33)13-12-19-15-21(38)11-10-20(19)16-32)27(41)34-17-23(39)35-22(14-18-8-6-5-7-9-18)26(40)36-25(28(42)43)31(3,4)46-45-30/h5-11,19-20,22,24-25H,12-17,33H2,1-4H3,(H,34,41)(H,35,39)(H,36,40)(H,37,44)(H,42,43)/t19?,20?,22-,24-,25-,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 24.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]-[p-Cl-Phe4]-DPDPE (delta opioid receptor selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase 1
(Leishmania major) | BDBM50600694

(CHEMBL5204239)Show SMILES Nc1nc(N)c2nc(CNc3ccc(cc3)C(=O)NCCc3ccccc3)cnc2n1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600691
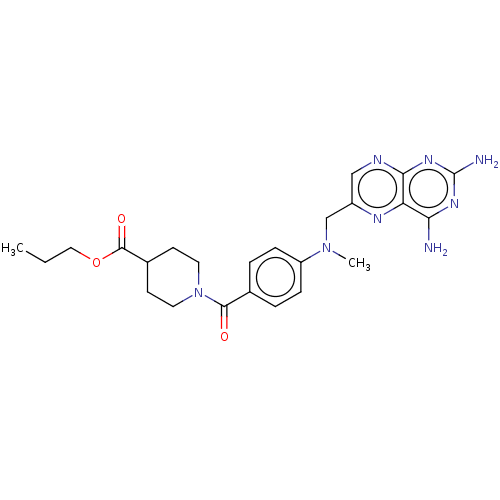
(CHEMBL5205559)Show SMILES CCCOC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(C)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase 1
(Leishmania major) | BDBM50600693

(CHEMBL5175012)Show SMILES Nc1nc(N)c2nc(CNc3ccc(cc3)C(=O)c3ccccc3)cnc2n1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50600679

(CHEMBL426 | Methotrexate | TCMDC-123832 | TCMDC-12...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600685

(CHEMBL5171609)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(CCCO)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Leishmania major) | BDBM50600679

(CHEMBL426 | Methotrexate | TCMDC-123832 | TCMDC-12...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600690

(CHEMBL5206994)Show SMILES CCOC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(C)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600682

(CHEMBL5193564)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(CC#C)Cc1cnc2nc(N)nc(N)c2n1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000807

(13-[2-Amino-3-(4-hydroxy-3-methoxy-phenyl)-propion...)Show SMILES COc1cc(C[C@H](N)C(=O)N[C@H]2C(=O)NCC(=O)N[C@@H](Cc3ccccc3)C(=O)N[C@@H](C(O)=O)C(C)(C)SSC2(C)C)ccc1O Show InChI InChI=1S/C31H41N5O8S2/c1-30(2)24(35-26(39)19(32)13-18-11-12-21(37)22(15-18)44-5)28(41)33-16-23(38)34-20(14-17-9-7-6-8-10-17)27(40)36-25(29(42)43)31(3,4)46-45-30/h6-12,15,19-20,24-25,37H,13-14,16,32H2,1-5H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t19-,20-,24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]-[p-Cl-Phe4]-DPDPE (opioid receptor delta selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase 1
(Leishmania major) | BDBM50600703

(CHEMBL5201183)Show SMILES CCN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)c1ccccc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50398394

(CHEMBL1232702)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(C)Cc1cnc2nc(N)nc(N)c2n1 Show InChI InChI=1S/C22H26N8O3/c1-29(12-15-11-25-19-17(26-15)18(23)27-22(24)28-19)16-5-3-13(4-6-16)20(31)30-9-7-14(8-10-30)21(32)33-2/h3-6,11,14H,7-10,12H2,1-2H3,(H4,23,24,25,27,28) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600679

(CHEMBL426 | Methotrexate | TCMDC-123832 | TCMDC-12...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000800

(13-[2-Amino-3-(4-hydroxy-3-iodo-phenyl)-propionyla...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)c(I)c2)C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O Show InChI InChI=1S/C30H38IN5O7S2/c1-29(2)23(35-25(39)19(32)13-17-10-11-21(37)18(31)12-17)27(41)33-15-22(38)34-20(14-16-8-6-5-7-9-16)26(40)36-24(28(42)43)30(3,4)45-44-29/h5-12,19-20,23-24,37H,13-15,32H2,1-4H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t19-,20-,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]- DPDPE (delta opioid receptor selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase 1
(Leishmania major) | BDBM50600684

(CHEMBL5182125)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(SCc2cnc3nc(N)nc(N)c3n2)nc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000799

(13-[2-Amino-3-(4-hydroxy-phenyl)-butyrylamino]-7-b...)Show SMILES C[C@@H]([C@H](N)C(=O)N[C@H]1C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](C(O)=O)C(C)(C)SSC1(C)C)c1ccc(O)cc1 Show InChI InChI=1S/C31H41N5O7S2/c1-17(19-11-13-20(37)14-12-19)23(32)27(40)35-24-28(41)33-16-22(38)34-21(15-18-9-7-6-8-10-18)26(39)36-25(29(42)43)31(4,5)45-44-30(24,2)3/h6-14,17,21,23-25,37H,15-16,32H2,1-5H3,(H,33,41)(H,34,38)(H,35,40)(H,36,39)(H,42,43)/t17-,21+,23+,24+,25+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 85 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]-[p-Cl-Phe4]-DPDPE (delta opioid receptor selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600681

(CHEMBL5180552)Show SMILES CCN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N1CCC(CC1)C(=O)OC | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000808

(13-[2-Amino-3-(3-amino-4-hydroxy-phenyl)-propionyl...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)c(N)c2)C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O Show InChI InChI=1S/C30H40N6O7S2/c1-29(2)23(35-25(39)19(32)13-17-10-11-21(37)18(31)12-17)27(41)33-15-22(38)34-20(14-16-8-6-5-7-9-16)26(40)36-24(28(42)43)30(3,4)45-44-29/h5-12,19-20,23-24,37H,13-15,31-32H2,1-4H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t19-,20-,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 93 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for its inhibitory potency against Opioid receptor delta 1 of Mouse vas deferens |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase 1
(Leishmania major) | BDBM50600702

(CHEMBL5199171)Show SMILES CCOC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(CC)Cc1cnc2nc(N)nc(N)c2n1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50398395
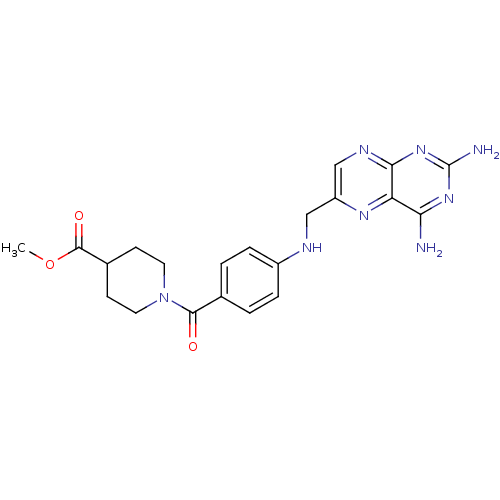
(CHEMBL1232399)Show SMILES COC(=O)C1CCN(CC1)C(=O)c1ccc(NCc2cnc3nc(N)nc(N)c3n2)cc1 Show InChI InChI=1S/C21H24N8O3/c1-32-20(31)13-6-8-29(9-7-13)19(30)12-2-4-14(5-3-12)24-10-15-11-25-18-16(26-15)17(22)27-21(23)28-18/h2-5,11,13,24H,6-10H2,1H3,(H4,22,23,25,27,28) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50000808

(13-[2-Amino-3-(3-amino-4-hydroxy-phenyl)-propionyl...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)c(N)c2)C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O Show InChI InChI=1S/C30H40N6O7S2/c1-29(2)23(35-25(39)19(32)13-17-10-11-21(37)18(31)12-17)27(41)33-15-22(38)34-20(14-16-8-6-5-7-9-16)26(40)36-24(28(42)43)30(3,4)45-44-29/h5-12,19-20,23-24,37H,13-15,31-32H2,1-4H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t19-,20-,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 111 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for its inhibitory potency against delta opioid receptors of Mouse vas deferens |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Pteridine reductase, putative
(Trypanosoma brucei brucei (strain 927/4 GUTat10.1)) | BDBM50600695

(CHEMBL5208403)Show SMILES COc1ccc(CNC(=O)c2ccc(CNCc3cnc4nc(N)nc(N)c4n3)cc2)cc1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Bifunctional dihydrofolate reductase-thymidylate synthase
(Leishmania major) | BDBM50600690

(CHEMBL5206994)Show SMILES CCOC(=O)C1CCN(CC1)C(=O)c1ccc(cc1)N(C)Cc1cnc2nc(N)nc(N)c2n1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00232
BindingDB Entry DOI: 10.7270/Q2CJ8JJC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50000807

(13-[2-Amino-3-(4-hydroxy-3-methoxy-phenyl)-propion...)Show SMILES COc1cc(C[C@H](N)C(=O)N[C@H]2C(=O)NCC(=O)N[C@@H](Cc3ccccc3)C(=O)N[C@@H](C(O)=O)C(C)(C)SSC2(C)C)ccc1O Show InChI InChI=1S/C31H41N5O8S2/c1-30(2)24(35-26(39)19(32)13-18-11-12-21(37)22(15-18)44-5)28(41)33-16-23(38)34-20(14-17-9-7-6-8-10-17)27(40)36-25(29(42)43)31(3,4)46-45-30/h6-12,15,19-20,24-25,37H,13-14,16,32H2,1-5H3,(H,33,41)(H,34,38)(H,35,39)(H,36,40)(H,42,43)/t19-,20-,24-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 137 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for its inhibitory potency against Opioid receptor delta 1 of Mouse vas deferens |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(MOUSE) | BDBM50000805

(13-[(2-Amino-6-hydroxy-1,2,3,4,4a,8a-hexahydro-nap...)Show SMILES CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@]2(N)CCC3CC(=O)C=CC3C2)C(=O)NCC(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@H]1C(O)=O |c:20| Show InChI InChI=1S/C32H43N5O7S2/c1-30(2)24(37-29(44)32(33)13-12-19-15-21(38)11-10-20(19)16-32)27(41)34-17-23(39)35-22(14-18-8-6-5-7-9-18)26(40)36-25(28(42)43)31(3,4)46-45-30/h5-11,19-20,22,24-25H,12-17,33H2,1-4H3,(H,34,41)(H,35,39)(H,36,40)(H,37,44)(H,42,43)/t19?,20?,22-,24-,25-,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 137 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Compound was evaluated for the binding affinity in comparison with [3H]-[p-Cl-Phe4]-DPDPE (opioid receptor delta selective ligand) |
J Med Chem 35: 2384-91 (1992)
BindingDB Entry DOI: 10.7270/Q2DF6Q4K |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data